Tagging system may make building biologically relevant proteins faster, cheaper and greener
Chemists in Germany have devised a faster, cheaper and greener way to synthesise proteins which avoids the need to use high pressure liquid chromatography (HPLC).
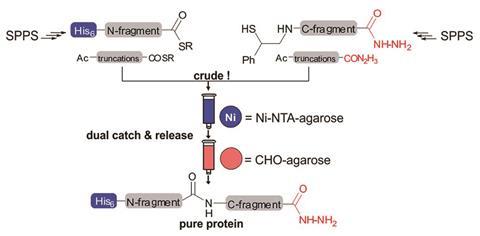
Being able to chemically synthesise proteins from scratch allows them to be modified with atom-by-atom precision, and in ways that are not possible in nature. For example, researchers can introduce non-natural amino acids into proteins or tag them with fluorescent markers.
Most biologically interesting proteins have in the region of 300 amino acid residues. These have to be made in smaller fragments which usually require purification by HPLC before they are added together to build the complete protein. HPLC is hugely time consuming, and some proteins can take months to make. It also requires expensive machinery and large volumes of organic solvents like acetonitrile.
Oliver Seitz and his team at Humboldt University of Berlin, Germany, have come up with a way to do in three days what used to take a month, by eliminating the need for HPLC. ‘In principle this should also be relatively easy to automate,’ says Seitz. ‘It involves only liquid dispensing and filtration. Any high throughput facility already has robotic dispensers that can do this anyway, and they could be reformatted for our protein synthesis method.’
Their method is based on a tagging system which weeds out incomplete protein fragments at each stage. For every step in their synthesis, they introduced chemical functionality to peptide ends that allowed the desired product to be easily identified. These functionalities come in the form of histidine and hydrazone tags which enable immobilisation on solid substrates, allowing other impurities to be washed away. They used a 2-mercapto-2-phenylethyl group to couple two fragments – adding it to the end of a fragment meant that only complete fragments could react.
Oleg Melnyk, Director of Research at the National Centre for Scientific Research in France, thinks that this is an important advance in protein synthesis. ‘It enables the synthesis of proteins in high purity, relying on robust and affordable solid phase techniques that can be potentially automated,’ he says.
‘At the moment we have only showed the coupling of two fragments because we studied proteins that are not very large,’ explains Seitz. ‘You would need several fragments for larger proteins – which should be feasible, but now this is what we must explore.’











No comments yet