Caging a single molecule of HF in a fullerene cavity could help researchers study its properties
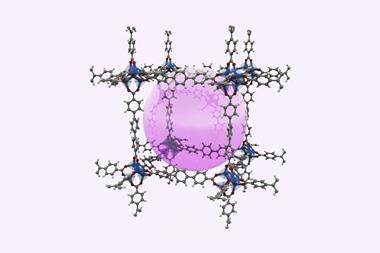
Researchers have used molecular surgery to trap a single hydrogen fluoride (HF) molecule inside a fullerene cage for the first time. HF normally exists as a dimer and so trapping an individual molecule could help scientists better understand its fundamental properties under conditions which cannot be achieved by studying the free, highly reactive and toxic molecule.
Other isolated molecules and atoms have previously been caged inside a fullerene cavity, including water, helium and hydrogen. While attempts have been made to trap a HF molecule, they resulted in unsymmetrical open-cage versions due to difficulties in closing the hole through which the HF molecule entered the fullerene. The lack of symmetry meant certain analyses were impossible while the hole allowed the HF to escape.
Now, a team led by Richard Whitby at the University of Southampton, UK, has found a way to chemically stitch up the fullerene hole and keep a HF molecule contained to produce a HF@C60 endofullerene. ‘The achievement of the synthesis was not surprising, just challenging,’ says Whitby. It took his PhD student Andrea Krachmalnicoff six months to find the right conditions for it to work, he adds.
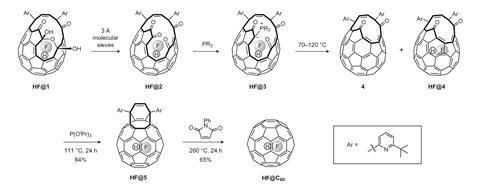
The easy part was making a hole in a C60 fullerene that was big enough for a molecule of HF to pass through. To do this, the team used a technique involving chemical transformations that was developed by Yasujiro Murata in 2011 to encapsulate water inside a fullerene. However, the team were met with a challenge when it came to closing the hole. The HF was lost under the closure conditions of Murata’s method, which involved heating with triphenylphosphine.
The key to closing the cage was to find an optimal phosphine for the job of converting a carbonyl group into a phosphorus ylide, which then undergoes a Wittig reaction to chemically stitch the hole up fast enough to prevent the HF escaping. Theoretical calculations of different phosphines by changing the groups attached to the phosphorus atom led the researchers to the ideal candidate of bis-2-furyl(phenyl)phosphine.
’There have been several computational studies to try to predict the properties of HF@C60 so it is nice to be able to test these against reality,’ says Whitby. ’The closed version has very high icosohedral symmetry which makes it possible to analyse the data on vibrational and rotational quantum states we obtain through IR and Inelastic neutron scattering data.’
‘The strategy in the successful optimisation process is quite remarkable,’ comments Liangbing Gan who investigates fullerenes at Peking University, China. However, he says it is a highly specialised protocol. ‘A more practical simplified synthetic procedure remains to be developed for such endohedral complexes in order for their easy access to those who are not so skilled synthetic chemists.’
References
A Krachmalnicoff et al, Nat. Chem., 2016, DOI: 10.1038/nchem.2563







No comments yet