A phosphorus(v) reagent system developed by US scientists can compete with the industry-standard phosphorus(iii) method for making oligonucleotide drugs, but offers chemists completely new possibilities for the types of molecules they can make.
‘We describe a new way to not only synthesise therapeutic oligonucleotides but also offer unprecedented ease in accessing a diverse array of chimeric oligonucleotides – with multiple phosphate linkages, installed interchangeably in a single synthetic platform,’ says Martin Eastgate from the pharmaceutical company Bristol Myers Squibb, who led the study alongside BMS colleagues and Scripps Research Institute’s Phil Baran.
Oligonucleotides are short DNA or RNA molecules that can be used in gene therapies against different diseases. There are hundreds of oligonucleotides in clinical trials and about a dozen are already used to treat disorders such as Duchenne muscular dystrophy.
Oligonucleotides are usually made through phosphoramidite chemistry – an approach that was developed in the 1980s and has been optimised and automatised since then. ‘Those methods revolutionised oligonucleotide synthesis and consequently oligonucleotide therapeutics,’ comments Punit Seth, who develops RNA-based drugs at Ionis Pharmaceuticals in the US. ‘They use trivalent phosphorus building blocks, which work great but are sensitive to water, and controlling chirality during synthesis can be challenging.’
Seth explains that replacing phosphorus(iii) with phosphorus(v) can reduce sensitivity to water, but phosphorus(v) building blocks are less reactive. ’The [researchers] use their recently developed phosphorous-sulfur incorporation reagents to enhance the reactivity of the phosphorus(v) building blocks,’ he says.
Maartje Bastings, a biomaterials expert from the Swiss Federal Institute of Technology, explains that natural DNA has phosphodiester bonds between the backbone sugars – the new approach allows researchers to create phosphorothioate connections instead. ‘This subtle difference has a great impact on the biostability of free synthetic DNA,’ she says.
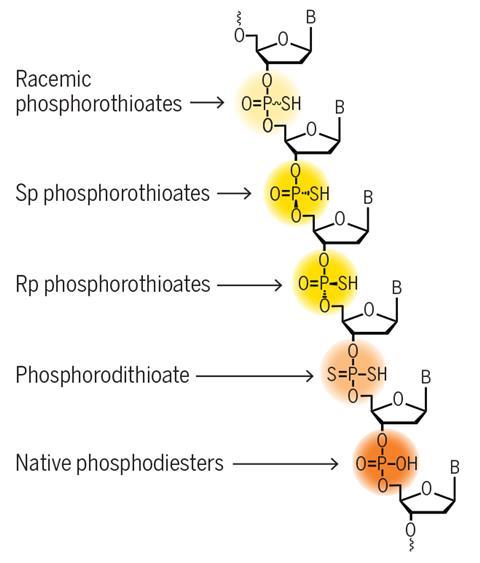
Eastgate’s BMS colleague Ivar McDonald says that installing such modified linkages is crucial to tune the properties of antisense oligonucleotides – molecules that interact with the mRNA of a target gene, influencing protein expression. ‘Traditional phosphoramidite chemistry prepares phosphorothioate linkages as mixtures of stereoisomers, whereas this method provides easy access to single isomers,’ he says. The synthesis can also create other types of linkages such as phosphodiesters, phosphorodithioates and racemic phosphorothioates in a single platform.
‘The reagents are prepared in very short sequences and many have green, renewable sources; for example, limonene from waste citrus peels,’ points out Eastgate’s BMS colleague Michael Schmidt. ‘The chemistry is also simple to carry out and the reagents are stable, crystalline solids, which aids greatly in handling and use.’
‘We have synthesised the reagents on kilo scales and are continuing to work with our partner, Millipore-Sigma, to make these reagents commercially available to the public,’ adds Schmidt.
The reaction is compatible with automated protocols and performs just as well as the industry standard phosphorus(iii) protocol, with all reactions reaching full conversion in less than two minutes. ‘… Our hope is that chemists will embrace [our methods] and find new opportunities that may not have been accessible using phosphorus(iii),’ McDonald says. ‘But we recognise that there won’t be an overnight change, as phosphorus(iii) methods and supply chains have been highly optimised and work very well for what they can do.’
References
Y Huang et al, Science, 2021 (DOI: 10.1126/science.abi9727)










No comments yet