Micromachines capable of navigating a tiny ‘lab on a chip’ have been developed by scientists in Spain. Protected by a protein-resistant layer, the micromotors can even swim through complex biological fluids, opening the door on a greater range of clinical devices for early diagnosis and analysis.
Micromotors have been used for a whole range of applications from mopping up bacteria to hydrogen generation. Now, research conducted by Alberto Escarpa, Beatriz Jurado-Sánchez and co-workers at the University of Alcalá, combines the merits of these tiny machines with microscale analysis of a lab-on-a-chip device. Controlled by a magnetic field, the micromachines can navigate their tiny lab, carry out fluorescence-based molecular detection and transport cargo in simulated blood plasma.
Normally, such lab-on-chip devices require tiny valves and pumps to precisely manipulate the sample, but these mechanisms are replaced by the rocket-like particles. The micromachines can move around in hydrogen peroxide solution by breaking down the peroxide over a platinum catalyst, producing a driving flow of oxygen bubbles.
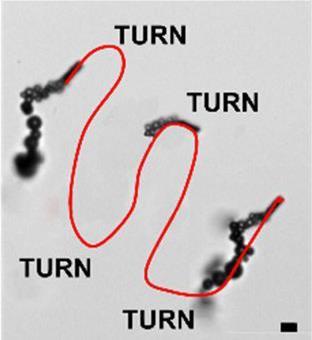
A key development in the reported magnetic carbon micromotors is their potential to be used in more complex media compared with previous works. This is thanks to a unique inner poly-O-phenylenediamine layer that prevents biofouling, the accumulation of proteins that can stop the platinum catalyst from working. Motors without this layer stop working when added to a protein-rich solution such as blood plasma, whereas the coated motors can effectively transport sugar-modified polystryene particles around the device.
Although the use of nanomachines have already been demonstrated by several groups over the past five years, Joseph Wang, a nanobioelectronics expert from the University of California San Diego, US, feels that Escarpa’s and Jurado-Sánchez’s work is ‘a wonderful example’ of using carbon-based microrockets for active transport.
This is backed by John Gibbs, who researches nanomotors at Northern Arizona University, US, who agrees that while the field has promised ground-breaking applications, ‘very few practical applications have thus far been achieved’. He calls this work is a ‘potential breakthrough’ especially with the development of the anti-fouling protective layer, which ‘overcomes a major limiting factor in the advancement of bio-medical applications in this field’.
In the future, Escarpa, Jurado-Sánchez and their team will be working in close collaboration with hospitals to develop a prototype for diagnosing sepsis in newborn children. ‘We hope that one day this new device will contribute to reduce the high mortality rates associated with this serious disease,’ says Escarpa.
References
This article is free to access until 14 June
R Maria-Hormigos, B Jurado-Sánchez and A Escarpa, Nanoscale, 2017, DOI: 10.1039/C6NR09750B

















No comments yet