An engineered nanoparticle has been created that can kill particularly aggressive cancer cells by starving them of copper. While this strategy has been attempted before, this is the first time researchers have used a copper depleting particle that does not appear to damage healthy cells. The team is confident that this could be the starting point for new, safer and more effective drugs.
‘Our goal was to develop novel nanomedicine that can be used against very aggressive, non-treatable triple negative breast cancer,’ says Jianghong Rao, who authored the paper. Triple negative breast cancer is rare and limited treatment options are available. These types of cancer cells grow and spread faster, and have worse outcomes than other types of invasive tumours.
A common feature of breast cancers is that they are more dependent on copper than healthy cells. As these aggressive triple negative cancers grow so fast they need a great deal of energy, which is supplied by mitochondria, cells’ powerhouses. Mitochondria cannot function without copper, as the element is vital for the electron transport chain that drives the production of ATP, the cell’s energy currency. Previous studies have showed that stopping copper trafficking disrupted energy production and slowed proliferation of cancerous cells.
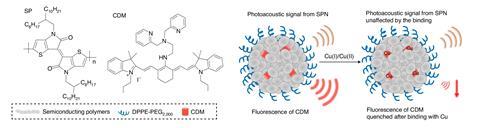
Rao’s team built their nanoparticle with three main features: a copper-depleting moiety that binds copper, a lipid coating to stabilise the particle and a polymer matrix with optical properties that makes it easy to detect in the body. The nanoparticle’s copper-depleting moiety is linked to a fluorescent dye and when copper binds it, the dye is quenched – something Rao calls a self-reporting ability.
The team then administered the nanoparticle to mice with cancer and analysed its impact. They found that the mitochondria in cancer cells treated with the nanoparticle were compromised. According to Rao, the nanoparticle did not affect healthy cells because they need less copper, so their uptake of the nanoparticle is lower too. The treatment downregulated key enzymes that deliver copper to mitochondria, interrupting copper trafficking and energy production. Even though cells can produce energy via other routes these were not enough to counter severe ATP deficiency. This energy shortage, coupled with compromised mitochondrial membrane potential and elevated oxidative stress, resulted in apoptosis. ‘Now we have a novel approach to targeting triple-negative breast cancer, both by exploiting its metabolism as well as by developing this novel drug delivery nanoparticle,’ Rao says. ‘Definitely, there is no limit to the application of this technology. Any cancer susceptible to mitochondrial inhibition may be treatable by our nanoparticles.’
Stephanie Blockhuys, who has worked on copper metabolism in human cancer cells, says the study is innovative. However, the postdoc at Cold Spring Harbor Laboratory says she is sceptical about its therapeutic potential. ‘The fundamental research on the copper-depleting nanoparticle is too limited to even think about clinical trials.’
Pernilla Stafshede, a biochemist at Chalmers University of Technology, adds that ‘the topic is timely – copper definitely plays a key role in cancer progression, and finding ways to limit cancer-promoting functions by cutting copper supply is a direction that should be further investigated.’ However, she adds that the study is missing ‘several pieces of information’ and that there needs to be better explanations and more data, before getting too enthusiastic. ‘If – and it’s a big if – this system truly targets cancer cells over other cells in an animal, and the proposed mode of action holds true, it may have great promise.’
References
L Cui et al, Nat. Biotechnol., 2020, DOI: 10.1038/s41587-020-0707-9










No comments yet