Meera Senthilingam
This week, we take on the snow - all thanks to this compound. Here's Phillip Broadwith:
Phillip Broadwith
A molecule that will be intimately familiar to anyone who has tried to scrape a thickly encrusted layer of ice from the windscreen of a car is ethylene glycol - the principal ingredient of antifreeze and de-icing sprays.
Spraying a solution of this simple molecule onto an icy surface can melt away the ice and allow you to get on with your journey. And mixing it with the water in a car's windscreen washing jets stops that from freezing too - so you can keep the windscreen as clear as when you set off.
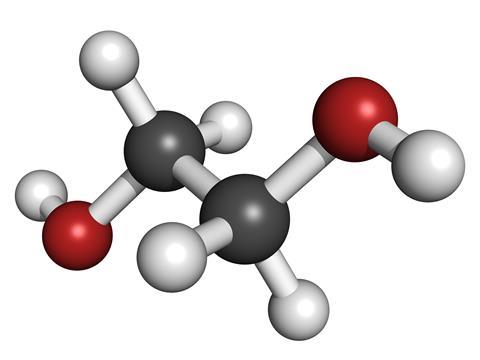
But how does it work? Ethylene glycol - or 1,2-ethanediol [NB. This is an error on the audio track] to give it its more systematic name - can do this because it is structurally quite similar to water. It comprises two linked carbon atoms, each bearing a hydroxyl - or OH - group. Because of these hydroxyl groups, it can form hydrogen bonds between molecules in the same way that water does. This makes it miscible with water in all proportions.
The freezing point of pure water, as we all know, is 0?C. And the freezing point of pure ethylene gycol is -12?C. But when the two are mixed together, they interfere with each other's attempts to organise into ordered solid structures. In fact this is quite a general phenomenon - dissolving a solute will usually depress the freezing point of the solvent and this is and this is exactly the same way that spreading salt on the surface of roads stops them from icing over.
So why don't we just use salt to deice our cars? Well, salt can only depress the freezing point of water to about -10?C - which might not be enough in the depths of winter to actually thaw the ice out. On the other hand, 70 per cent ethylene glycol in water won't freeze until about -50?C. Salt is also rather corrosive to metal car parts, which ethylene glycol is not.
However, there is a down side to the popularity of ethylene glycol as an antifreeze, and that is its toxicity. Pure ethylene glycol tastes and smells quite sweet, and this means that pets such as cats or dogs will quite often lap up spillages and become very ill. For this reason most commercial antifreeze products now contain very bitter additives to deter people and animals form drinking them.

This sweetness is also the root of a common misconception that antifreeze is added to wine by unscrupulous winemakers to make it sweeter. In fact, these villains are more likely to add diethylene glycol - two molecules of ethylene glycol joined together by an ether (or C-O-C) link - which retains the sweetness but is much less toxic.
If you or your pet are unlucky enough to drink antifreeze by accident, then it is important to get medical attention very quickly. Because of its similar structure to ethanol (or alcohol) it initially makes you very drunk very quickly, but also makes you nauseous and likely to be sick. As the body tries to metabolise the toxin, its hydroxyl groups are oxidised to carboxylic acids. This spreads the effect into the nervous system and can cause increased heart rate, high blood pressure and even heart failure. After around 24 hours, the kidneys will begin to fail under the strain.
Happily, treatment for ethylene glycol poisoning is relatively straightforward if caught early on. The enzyme that is responsible for converting ethylene glycol into its toxic metabilites is the same one that metabolises alcohol, but its affinity for alcohol is 100 times higher. Therefore a large dose of alcohol - usually given by intravenous drip rather than a good slug of whisky - can saturate the enzyme, preventing it from converting the glycol into more toxic compounds. This may stop the poisoning, but my guess is that you'd end up with the mother of all hangovers the next morning.

But antifreeze only accounts for a small percentage of the 20 million or so tonnes of ethylene glycol produced every year. The vast majority of this simple diol is used to make polyester plastics, particularly poly(ethylene terephthalate) or PET - the transparent, shiny and easily processed plastic that makes drinks bottles and fibres for clothing and textiles.
So the next time you wrap up warm in a fleecy jumper made from polyester fibres to spray and scrape the ice from your car windscreen, spare a thought for the little molecule that makes it all possible.
Meera Senthilingam
Indeed, the little molecule that certainly makes things more bearable in the winter. That was Chemistry World's Phillip Broadwith, with the sweet yet toxic chemistry of ethylene glycol. Now, next week, a way to bring in the New Year with a bang.
Brian Clegg
Nitrogen triiodide is a touch sensitive explosive. The lightest pressure and it blasts out of existence. In anything more than a pinch this is a dangerous substance, but in a thin coating it produces entertaining cracks and bangs.
This was a wonder substance to a typical teenager. A very thin coating on floor tiles produced a highly satisfying booby trap of a room that couldn't be crossed without a dance of shock as the floor erupted in a series of minor explosions.
Meera Senthilingam
And to find out more about the explosive chemistry of nitrogen triiodide, join Brian Clegg in next week's Chemistry in its element. Until then, thank you for listening. I'm Meera Senthilingam.













No comments yet