Meera Senthilingam
This week, Jen Newton reveals her arch enemy and the compound that helps her fight it.
Jen Newton
Ground elder is my nemesis. No matter how much time I spend weeding this thug of a plant, it always comes back. So I’m driven to reach for the Roundup. I’m in good company for Roundup is the world’s bestselling weedkiller.

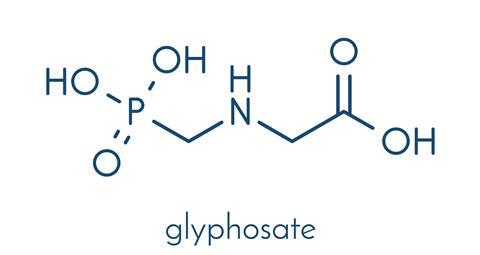
Roundup’s active ingredient is glyphosate. Its herbicidal activity was discovered by John Franz in 1970 while working for agricultural biotechnology company Monsanto, based Missouri in the US. The chemical itself was discovered 20 years earlier by Swiss chemist, Henri Martin, at the pharmaceutical company Cilag.
Glyphosate’s weed killing activity only came to light when Monstanto scientists were testing different compounds as potential water-softening agents. Two molecules closely related to glyphosate were found to have some herbicidal activity against perennial weeds so Franz then synthesised derivatives of those two compounds and quickly discovered glyphosate to be a potent herbicide. Monsanto patented glyphosate in the 1970s and retained exclusive rights to its use until the patent expired in September 2000.
The discovery of glyphosate had an incredible effect on the agricultural community, allowing farmers, as well as anyone with a garden, to easily and effectively control the growth of weeds. A huge number of glyphosate containing herbicides are now available. In fact, there are over 750 products containing glyphosate for sale in the US. It’s used in all sorts of places – agriculture, forestry, lawns and gardens, even products for controlling aquatic plants contain glyphosate. It is also used extensively in controversial programmes to eradicate drug producing plants such as coca, opium poppies and marijuana. As a non-selective weed killer, it will kill any plant it touches, and many gardeners will tell you how they have noticed mysterious yellow footprints across their lawn, only to realise they must have accidentally gotten weed killer on their shoes.

A condensation reaction known as the Mannich process is the most widely used and most widely developed method for its synthesis. On its own, glyphosate is not water soluble enough to be easily sprayed and taken up by the plant, so in many commercial products it is found as a soluable salt, often an ammonium salt or an isopropyl amine salt.
Glyphosate imparts its lethal effect on plants by inhibiting an enzyme called 5-enolpyruvylshikimate-3-phosphate synthase, or EPSP synthase for short. This enzyme is central to the penultimate step in the shikimate pathway, which is essential for the biosynthesis of aromatic amino acids like tryptophan, tyrosine and phenylalanine. A third of plant matter is estimated to consist of aromatic compounds derived from shikimate so it is easy to see why interfering with this vital biosynthetic pathway is a death sentence to plants.
The shikimate pathway is present in plants and bacteria but absent in mammals meaning humans can safely use glyphosate and eat plants that have been treated with it. Having said that, it is toxic in acute high doses, so it is still important to take the necessary precautions detailed on the bottle.
Glyphosate weed killers are applied directly to visible weeds. It is translocated via the plant’s phloem to meristematic tissue, underground storage organs and stem apicies. By disrupting such an important pathway in a wide range of plant tissues, glyphosate is able to control established perennial weeds as well as annual weeds.
Monsanto have since created glyphosate-resistant crops, so-called Roundup-Ready varieties of commercial crops, by inserting a gene derived from a petunia that produces large amounts of EPSP synthase into plants like soybeans, cotton, canola, and corn. The modified crops can produce enough EPSP synthase to overcome the inhibition that glyphosate causes, allowing farmers to spray Roundup directly on their fields, thus destroying every weed and plant except their Roundup-Ready crop. Since glyphosate-resistant crops offer the promise of a cheaper and simpler weed management process, farmers have adopted these crops at such an alarming rate that they now cover over tens of millions of acres worldwide.
So glycosphate and engineered crops have allowed for a dramatic change in farming practices, but not without some concerns. Very few studies have tested whether cross pollination between transgenic glyphosate resistant crops and their weedy relatives may result in super weeds, which could become rampant in a field of otherwise monocultured GM crops. Research is also underway to ascertain the effect of glyphosate on human health, as although its toxicity is well known, there is some concern about disruption of particular hormone systems. And, as with anything that can change our landscape so dramatically, research is ongoing into the true impact on the wider environment.
Meera Senthilingam
Chemistry World’s Jen Newton there, with the lethal chemistry of glyphosate. Next week, an unappreciated compound.
Brian Clegg
Many simple inorganic compounds, such as sodium chloride or iron oxide are familiar enough, but boron nitride – just one boron atom to one of nitrogen – has far less visibility than it deserves. It is a superbly useful compound, perhaps the best kept secret in all of chemistry.
Meera Senthilingam
Find out why by joining Brian Clegg in next week’s Chemistry in its Element. Until then, thank you for listening, I’m Meera Senthilingam.












No comments yet