Meera Senthilingam
This week, Kiki Sanford uses chemistry to highlight a useful life skill.
Kiki Sanford
Life is balance. A fine-tuning of processes at the molecular level that allow an organism to either remain alive or slip from this mortal coil into death.
But, how do things that are alive keep from tipping toward death? How do they maintain that perfect equilibrium?
The mechanisms that allow the littlest yeast cell to the largest elephant to carry on living are many, but arguably the actions of one small, nearly ubiquitous molecule appropriately called Ubiquitin have the most widespread impact of all. To be sure, cellular disruptions related to ubiquitin are involved in a variety of human medical disorders like cancer, neurodegeneration, muscle wasting, viral infections, diabetes and inflammation.
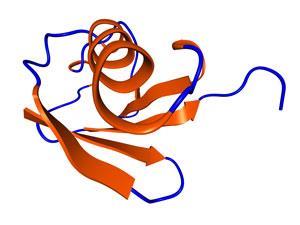
First identified in 1975 by a team of scientists at New York University led by Gideon Goldstein, ubiquitin was initially called ubiquitous immunopoetic polypeptide for its role in immune cell differentiation. However, because they found it everywhere they looked from cow to celery cells, the researchers recognized that the polypeptide chain weighing in at 8.5 kiloDaltons might play a much greater role in the cellular functions of all living organisms. It wouldn’t be tied to any of these until the publication of two papers in the Proceedings of the National Academy of Sciences by Aaron Ciechanover, Avram Hershko, and Irwin Rose in 1980.
In a 2006 interview, Ciechanover, a Nobel laureate and biochemist, said, ‘Life is a balance between division, growth, and no growth. Mutations can lead to imbalance unless repaired. Ubiquitin is involved in control of repair and in keeping balance.’
And, it is true. Even though the tiny molecule called ubiquitin is referred to time and again as the cellular ‘kiss of death’, its purpose is much greater. Ubiquitin explains how some proteins in cells are long-lived while others have only ephemeral intracellular existences, why some proteins flood cell interiors, and sightings of others are exceedingly rare. As a protein modifier, it really lies at the heart of how life persists against all odds by increasing the variety of ways cells and organisms can respond to their environment.
The most prominent role of this 76 amino acid compound is where its nickname derives – ubiquitin is a tag for proteins that require destruction or recycling. Hence, the ‘kiss of death’. Proteins labeled with chains of ubiquitin are broken apart through a process called proteolysis in which peptide bonds are destroyed, one by one.
It’s a fairly straight-forward process that was described by Hershko, Ciechanover (who was a graduate student at the time), and Rose. They elucidated the process now known as ubiquitination or ubiquitinylation, by which proteins are tagged with ubiquitin.
Between 1978 and 1983, the researchers discovered that ubiquitination is powered by ATP, which allows an enzyme called E1 to activate ubiquitin, a second enzyme called E2 conjugates the ubiquitin-E1 complex, which allows a third creatively titled enzyme called E3 to link ubiquitin to a target protein. In the final step, E3 is the molecule responsible for recognizing the substrate protein and catalyzing the ligation reaction.
There are literally hundreds of E3 molecules available to target specific proteins in the cell where and when needed. Organisms also have 16-35 E2 enzymes. But there is only one ubiquitin. The simple elegance of this hierarchical protein-modification system belies its importance.
How does ubiquitin binding possibly have the specificity to regulate the thousands of proteins that are involved with just about every cellular process from protein degradation to the cell cycle, transmembrane receptor trafficking, DNA repair, gene transcription, apoptosis, and more?
Well, this is where things really start to get interesting. It appears that the number of ubiquitin molecules in a chain dictate the fate of the tagged protein. So, the process of ubiquitination sometimes stops at the end of just one cycle. This is called monoubiquitination. Sometimes the process repeats again and again resulting in a chain that includes multiple ubiquitin moieties, all linked by an isopeptide bond between the C-terminus of the next ubiquitin in line and a lysine contained in the previous molecule.
Ubiquitin contains seven lysine residues, called K6, K11, K27, K29, K33, K48, and K63 because of their locations in the molecule. This allows a certain amount of flexibility to expand the tagging function of ubiquitin. For example, chains linked via the K48 residue are responsible for sending proteins for degradation. Other linkages, such as K63, might be involved in other processes, like DNA repair.
The current hypothesis is that the ubiquitin chain length and linkage type confer a specific topology to the complex that gets created with the target protein. This then determines how other molecules interact with it. For instance, it is possible that longer chains remain in the proteasome longer allowing more complete degradation.
As you can see, we now know that there are multiple layers to the ubiquitin protein modification system wherein specificity can be conferred. It’s hard to believe that just 40 years ago, we had no idea what ubiquitin was.
Prior to the 1950’s, cellular proteins were considered stable things that did particular jobs in the body without requiring much, if any, maintenance. Then, in 1942, work by Rudolf Schoenheimer at Columbia University elucidated the concept of the body, cells, and their constituent proteins as part of a dynamic system requiring constant change. Flux rather than stability became the norm.
How does a cell deal with the constant ebb and flow of internal and external forces, and break down proteins when it needs to clean house?
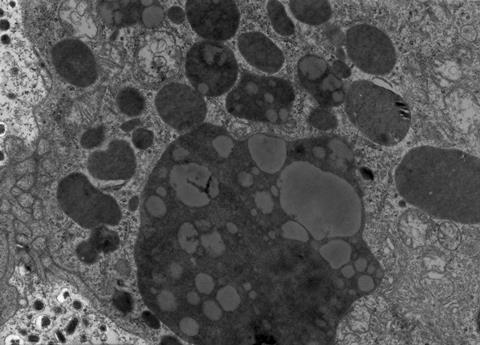
Lysosomes were discovered in the 1950’s by Christian de Duve, and provided a route for cellular junk disposal. However, they didn’t fit the bill entirely. In fact, lysosomal degradation of proteins could not explain: why different proteins had varied half-lives in cells; why degradation seemed to be linked to changes in physiological parameters like acidity, nutrient availability, or cellular growth phase; nor why lysosomal inhibition didn’t seem to affect some populations of proteins at all.
The most crucial component of protein degradation that the lysosome hypothesis could not address was the energy requirement that kept popping up. Digestive enzymes found in the lysosome don’t require any additional energy to lyse proteins. Yet, very often in the intracellular matrix ATP was needed to complete the breakdown process.
Work on a molecule called APF1, or alternately ATP-dependent proteolysis factor-1, by Hershko bucked the popular science trend during the 70’s of studying how proteins are formed, but was inspired by these somewhat neglected and anomalous findings of ATP dependency in protein degradation. APF1 was later identified as ubiquitin, and Ciechanover, Hershko, and Rose, who passed away in August 2015, were awarded the 2004 Nobel prize in chemistry for the work they undertook in the 1970’s and 80’s to uncover ATP-dependent ‘ubiquitin-mediated protein degradation’.
At the time they had no idea how important their work was going to be to our understanding of cell biology. Years later, we have made immense progress, but still we have only just begun to scratch the surface of ubiquitin, and how this fascinating system is controlled and in turn controls the balance of life.
Meera Sethilingam
Sceince writer Kiki Sanford with the regulating chemistry of ubiquitin. Now next week, we defy the rules of chemistry.
Kat Arney
Because high temperatures affect the structures of vital biological molecules like DNA and proteins, researchers had thought the upper limit for the survival of even the hardiest of bacteria was about 73°C. The man who challenged the idea life couldn’t take the heat was biologist, Thomas Brock.
Meera Sethilingam
Find out the result in next week’s Chemistry in its Element. Until then, thank you for listening. I’m Meera Sethilingam.













No comments yet