Sensor will continuously monitor drug concentrations in real time letting doctors tailor treatments to the patient
A sensor that can continuously monitor the concentration of a drug in the bloodstream is set to help personalised medicine take off. This technology will let doctors tailor drug treatment courses for each patient with exceptional precision and according to how fast they excrete or metabolise different medicines.
Continuous, real time monitoring of specific molecules in patients’ bloodstreams is already possible in a few special cases, most notably for glucose, where it has revolutionised the treatment of unstable diabetes. However, these systems are based on enzymes and their highly specific recognition of natural substances such as glucose or lactose. Unfortunately, drug targets are normally membrane proteins, which are notoriously unstable when removed from the cell. This means they can’t be integrated into a sensor to monitor what’s happening to a drug in the body.
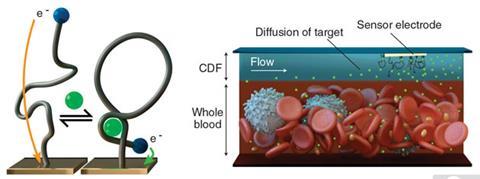
Tom Soh and his team at the University of California, Santa Barbara, US, got around this problem by using electronic aptamer sensors developed by the group of co-author Kevin Plaxco, also at Santa Barbara. DNA aptamers are strands of DNA that fold up into specific, protein-like shapes. By systematic selection from large pools of random DNA sequences, researchers can identify aptamers that recognise certain molecules very precisely, including both proteins and small molecule drugs. Once aptamer sequences that bind to the target of interest have been found they can be easily manufactured to order.
Sensors shape up
Plaxco’s group has developed electronic sensors that can pick up the large shape changes that aptamers can undergo when switching between their unbound state to when they are bound to their target. They employ this conformational change to enhance or suppress electron transfer between a redox reporter group attached to the DNA and an electrode to which the DNA is tethered. The researchers have previously shown that these sensors are cheap and easy to build when they created an extremely sensitive test for cocaine. The aptamer sensors are also robust enough to employ in complex ‘dirty’ samples like human blood serum and don’t need special reagents or washes so can be monitoring a sample constantly.
‘The sensors fail, though, when challenged directly in whole blood, which hampers their ability to measure drug concentrations in blood on-the-fly,’ Plaxco explains. ‘When one of my students described this problem in a joint group meeting between Soh’s team and ours, however, the engineers saw a solution to the blood-induced fouling problem that had long stymied our efforts.’
To make the sensor work with blood in real time, Scott Ferguson, a postdoc in the Soh group, added two new features. First, to stop the sensors from becoming clogged up with larger things like red blood cells, he developed a liquid barrier in the form of a thin film of buffer solution flowing between the sensors and the blood sample. With their higher mobility, small drug molecules easily move across this filter, while cells, which are much larger and thus diffuse much more slowly, are kept away from the sensor. ‘His second innovation was to probe the electrochemical sensors using potential pulses of various frequencies, which provides a means of correcting for any residual background drift, since the signals obtained at different frequencies drift in concert,’ Plaxco explains.
MEDIC!
To demonstrate the potential of the system, which they call MEDIC – Microfluidic Electrochemical Detector for In vivo Continuous monitoring – the scientists built a sensor to track blood concentrations of the cancer drug doxorubicin. This cancer drug wasn’t chosen by chance – it is notoriously difficult for doctors to administer patients with the optimum dose.
Using anaesthetised rats, the researchers showed that the device could monitor doxorubicin in the bloodstream. The sensor only needed a quarter of a millilitre of blood per hour, so could remain in operation for as long as is required. The authors have also tested the device in vitro on human blood, where it performed equally well. Clinical tests on human patients have yet to be carried out, however. Nevertheless, there are already specific aptamers for around a dozen pharmaceutically relevant molecules whose concentrations in the blood could be monitored by MEDIC.
Robin Felder, associate director of clinical chemistry at the University of Virginia, US, welcomes the advance. Looking ahead, Felder anticipates that ‘aptamer technology promises to revolutionise the scope of continuous clinical analysis in personalised medicine. Wearable sensors will report critical health issues on a minute-to-minute basis allowing for therapies that are tailored to the individual and their personal challenges with health.’
When introduced to the clinic, the device should be as versatile as the folding patterns of DNA. ‘Aptamers have a number of advantages for such measurements,’ says Ingolf Schimke from the Charité Hospital, Germany. ‘Aptamers are chemically synthetised and therefore can be readily standardised. Some of the aptamers are per se chemically stable in blood, other can be modified for increased stability. The crucial limitation is in the selection of new aptamers for specific targets.’











No comments yet