Researchers are on a quest to outsmart and overcome the sophisticated security system of the brain. Julia Robinson reports on some of the approaches being studied
Our brains are composed of billions of neurons that need constant protection from disease-causing pathogens and toxins circulating in the blood. Part of this protection is provided by the blood–brain barrier (BBB) – a highly selective, semipermeable border of endothelial cells which, as the name suggests, acts as an essential barrier between the brain’s blood vessels and other components that make up brain tissue.
‘If you were to take a blue dye and inject it into your body, everything would turn blue, but your brain would [remain] pristine white – the blood–brain barrier is very strict,’ says James Choi, a bioengineer at Imperial College London in the UK. Grateful as we are for this in-built security system, however, when things go wrong with the brain the BBB’s tough selection criteria presents a major obstacle.
‘The [blood–brain] barrier is essential to protect your brain, but when you have a disease it’s kind of a double-edged sword,’ explains Kevin Boyé, a junior group leader at Paris Cardiovascular Research Centre in France. ‘It’s your worst enemy, because it will prevent delivery of molecules to treat your disease.’ It is understood that only lipid-soluble molecules smaller than 400–500 Da can naturally cross the BBB, which excludes 98% of drugs.
For years it has been the goal of neuroscientists and vascular biologists to find a magic bullet for temporarily opening and resealing the BBB to administer much-needed treatments for conditions such as Alzheimer’s and brain cancer. Several breakthroughs have been made and we now have numerous potential strategies for crossing the BBB that have brought us closer to this goal. However, there is still much to learn about this elusive wall and how to successfully, and safely, cross it.
Understanding the barrier better
Over the past decade, scientists have developed more of a detailed understanding of how the BBB functions and the variety of signalling pathways that regulate its properties. ‘We know that we have a layer of endothelial cells that express a lot of tight junction [proteins] that seal the endothelial cells to one another,’ explains Boyé. ‘They also have a low rate of transcytosis so that will limit transcellular permeability. One of the very famous signalling pathways is Wnt signalling – we know that when you don’t regulate Wnt signalling, you will have defective tight junction expression and increased transcellular permeability,’ he adds.
Research has also shown that the BBB is not simply a barrier. Vivianna Gradinaru, a neuroscientist and biological engineer at the California Institute of Technology in the US, says the term is almost a misnomer. ‘It is not a monolithic, unchangeable homogeneous wall,’ she says. ‘It is a living, breathing entity, that is there to serve us in protecting us from circulating agents, whether it’s viruses or bacteria, but at the same time, is there to allow selective entry, or efflux of, immune protection, for example.’
These discoveries have led researchers to learn how different disease states can affect the BBB and enabled a rethinking of how to target it. ‘Now that we have new technologies like lipid nanoparticles and mRNA, people are thinking about different strategies in addition to crossing it; can we now hit the barrier itself and make it a local therapeutic depot?’ asks Michael Mitchell, a bioengineer at the University of Pennsylvania in the US.
Ultrasound and microbubbles
The premise of Choi’s work at Imperial is to develop a non-invasive method of opening the BBB using a novel ultrasound and microbubble approach to temporarily disrupt the barrier to enable drugs to enter the brain. Microbubbles are administered into the bloodstream which, in the presence of an acoustic field provided by transcranial focused ultrasound, start oscillating, stimulating the opening of the BBB. ‘You have these bubbles going through the entire body, including the brain and then you apply a very specific ultrasound sequence, which the bubbles will respond to,’ Choi explains. ‘We’re effectively creating a remote-control system for these bubbles in the brain’s blood vessels. We’re trying to control the oscillations very gently so that the blood–brain barrier can open up and then the drugs, which are already in the bloodstream, can come out into the brain region.’
The exact mechanism behind this is not yet fully understood but, as Choi explains, there are two leading theories. The first, more direct path, is that the method enables an ‘unzipping’ of the blood vessel which allows the drugs to go across before they zip up again. The second is that the method is mechanically stimulating the vessels, and this massaging action causes the cells to undergo transcytosis. ‘What that means is that the cells will fold into itself in certain parts of it – forming very small pockets – and these will act like vesicles, creating a cargo transportation system where the drugs can go across to the other side,’ says Choi.
It’s a game of ratios – how much do you need to put into the blood relative to how much gets into the brain
There are currently over 25 clinical trials, the majority phase 1, investigating the administration of a variety of drugs across the BBB using this approach, including the chemotherapy agents carboplatin and cisplatin, as well as panobinostat for paediatric brain cancer. Applications also include debilitating neurological conditions such as Rett syndrome and Parkinson’s, Huntington’s and Alzheimer’s diseases.
Choi is optimistic it will only be two to three years until this approach will be used in patients, but before then it will be necessary to show that enough drug can be delivered to have a tangible clinical impact. There are also potential risks to consider when opening this essential barrier, although Choi says that in his lab the opening is less than 10 minutes. ‘You have a very short window where you can inject a bolus of the drug and then it’ll close right back up,’ he says.
A major advantage of the microbubble/ultrasound approach is that you don’t need to change the design of existing drugs, meaning there is chance that drugs that have failed clinically in the past due to dose-associated side effects could be given a new lease of life. ‘It’s a game of ratios – how much do you need to put into the blood relative to how much gets into the brain,’ explains Choi. ‘If we can put more into the brain and less into the blood then the chances of the drugs being successful in a clinical trial increases.’
Viral vectors
When Gradinaru’s lab started their work on viral vectors back in 2011 there was limited understanding of the BBB and its biology. And only a small number of targets, such as the transferrin receptor, a membrane receptor involved in the control of iron supply to the cell through the binding of transferrin, have been validated for research and therapies. But just over a decade after they started, Gradinaru’s team have successfully identified a previously unknown mechanism by which certain gene delivery vectors, such as adeno-associated viruses (AAVs) can cross through the BBB.
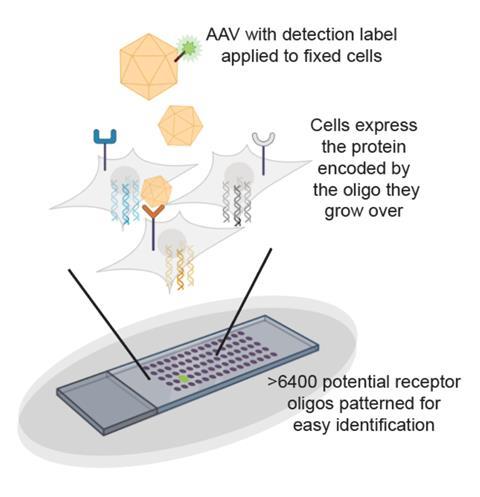
To make this discovery her group turned to directed evolution to guide the development of a panel of AAV vectors and enhance their ability to cross the BBB from the bloodstream. Using this method they have been successful in identifying AAVs that cross the BBB in different strains of mice, rats, marmosets and in the macaque. ‘Because our screens were very efficient and fast, by the time we got to non-human primates, we were able to get that in only two rounds of evolution with very little experimentation and very little loss,’ says Gradinaru.
However, they also found there were many other classes of molecules that could enable BBB transport. Two of these candidates – carbonic anhydrase IV (CA-IV) and low-density-lipoprotein-receptor-related-protein 6 (LRP6) – were of particular interest. ‘CA-IV is a really interesting finding, because it’s so unintuitive – it’s an enzyme and it’s very strongly present on the blood–brain barrier…it’s involved in pH balance,’ she explains.
‘CA-IV is very strong on the BBB but you don’t have it on the brain cells. LRP6 [is] both on the BBB and the brain cells,’ says Gradinaru. ‘What does this mean? If you want to be ubiquitous and strong, LRP6 is the best choice, if you want to have certain specificity, you go with CA-IV to cross and then maybe with a specific antibody or modality, to lock into the cell type.’
The antibody approach
The development and maintenance of the BBB are dependent on the Wnt signalling pathway, which regulates several crucial cellular processes. In 2022, Anne Eichmann’s team at Yale School of Medicine in the US successfully created an antibody that could block a specific ligand, called Netrin-1, from binding to its receptor. By doing this, the team were able to disrupt the Wnt signalling pathway, transiently opening the BBB to enable drug delivery.
Eichmann said they initially set out to answer just a ‘basic science question’. ‘We had made mouse models that disrupted either the receptor on endothelial cells or the ligand on all cells. So we made these knockout mice, they both had the same phenotype, and they both displayed an irreversibly leaky blood–brain barrier.
‘So our question was, if we selectively block the binding of this ligand to this receptor – because they don’t have a mutually exclusive relationship – we reasoned this should also give us a blood–brain barrier defect, but that in this case, the barrier defect should be reversible, meaning as soon as the antibody is cleared out of the circulation, the barrier would close again, and that’s exactly what we observed.’
We haven’t seen any toxic effects so far in our clinical models
This isn’t the first time that researchers have tried to target the Wnt pathway directly. Others have tried to engineer Wnt ligands or inhibitors to open the barrier but the problem with this strategy is that these Wnt molecules are made by astrocytes within the brain so they will induce an opening of the barrier at a site that is not in contact with the blood.
‘Our approach is different because our blocking antibody can be injected intravenously – we can open the barrier … via direct intravenous injection [which] makes it more translatable for human application,’ says Boyé, who led the research.
The finding gives rise to some interesting therapeutic possibilities and Eichmann’s team are attempting to deliver different kinds of drugs into mice disease models to test their efficacy. The approach is applicable to a large range of diseases including brain cancers, neurodegenerative diseases – such as Alzheimer’s – and various other diseases.
It will be necessary to assess the toxicity of the approach, however, and conduct a proof-of-concept study in rodents and then larger animals before moving to humans. ‘What we can say is that since we have a transient opening for a short period of time – five to six hours – we haven’t seen any toxic effects so far in our clinical models,’ explains Boyé. ‘It’s a question of balance of advantages and risks; when you have a brain tumour, survival is about 15 months, maybe you hope to take a therapy that will destroy your tumour but might lead to some toxicity.’
Novel nanoparticles
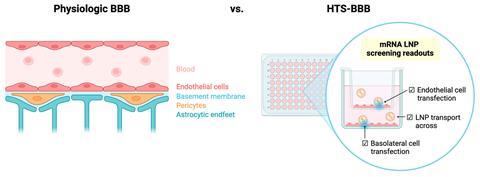
Another potentially safer BBB delivery vehicle can be found in lipid nanoparticles (LNPs) which can pass through the BBB and carry proteins, antibodies or even messenger RNA (mRNA) with them. The approach is being explored by a team at the University of Pennsylvania in the US, led by Michael Mitchell. There they have put together a simple model of the BBB allowing them to test the best drug transporters in the lab.
‘There are a number of different types of in vitro BBB models,’ explains Emily Han, a bioengineering PhD student at the University of Pennsylvania and lead author of this work. ‘The preferred ones for drug transport are transwell [systems] because they have that top and bottom compartment so you can put your nanoparticles – your drugs – into the top compartment and then see how many can cross the endothelial BBB layer and then enter the bottom compartment – which represents the brain.’
‘While most transwells only look at transport of the LNP into the bottom compartment, in this model, we can look at how many LNPs were trapped in the BBB and ended up just transfecting the BBB cells.’ She explains that LNPs are often a preferred delivery vehicle because they are so safe.
‘While viral vectors are often efficacious, it’s housed in a viral capsid so there’s a risk of that integrating into the genome and causing toxicity. LNPs, on the other hand, are made with lipid components that are often found naturally – so they’re really safe – and they can easily fuse with cell membranes, which we think helps them be taken up into cells.
One of the key components of an LNP, she adds, is the ionisable lipid. When it gets taken into the endosome of a cell, the lipid changes from neutral to positively charged and it can escape the endosome and the cargo can be more efficiently released into the cell.
Any kind of nucleic acid can be transported across the BBB using this method and this versatility means it can also be used to treat any kind of brain disease, as long as there is a protein that can be up- or down-regulated and used as a target for the LNP.
‘It can also be for any brain cell type, so for example, in cases of stroke, haemorrhage or traumatic brain injury, immediately the BBB is damaged, so there’s a good amount of research in trying to deliver LNPs to the BBB itself to try to repair the BBB or reduce inflammation,’ says Han.
Questions remain on how the LNPs exit the BBB and also where the majority of the package being delivered actually ends up ̶ as surprisingly little will actually make its way into the brain, even when the procedure has gone well. ‘When we think about brain delivery, the first thing we think about is can we get anything there, it’s so tough to do, even if we get 1% or 2% of the RNA we inject, that’s very exciting,’ says Mitchell.
Breaking the barrier
There is great optimism among the research community about finally being able to break through the blood–brain barrier – to conduct important studies and provide desperately needed treatments. But the excitement has been slow to manifest elsewhere which has created challenges for research in this area.
Currently there are not a lot of pharmaceutical companies that have a BBB programme and there may still be some work to do to persuade them of the value of investing in this area. Although Mitchell feels that the hardest to convince are funding agencies. ‘Some advice I got when I started my career as a professor is do not make your whole lab about the blood–brain barrier. It’s a career killer – it’s very high risk, high reward,’ explains Mitchell. ‘It can be very hard to get NIH [National Institutes of Health] funding. The opposite has been true for pharmaceutical companies and foundations – the ALS Foundation, the Rett syndrome foundation – fingers crossed we’ll be getting funding from them.’
In the UK, Choi says he reckons all the pharmaceutical companies are watching the ultrasound space to see if any of the methods being explored will pass the test and be used consistently. ‘I think for brain cancer, it’s very likely,’ he says. ‘The idea of having these non-invasive surgical procedures come about into the real world … it will happen, I just think it will take some time. One day we’ll never have to cut anyone open – that’s my dream.’
Julia Robinson is a science correspondent for Chemistry World















No comments yet