Comprehensive solvent acidity scale could help make acid-catalysed reactions more reliable and reproducible
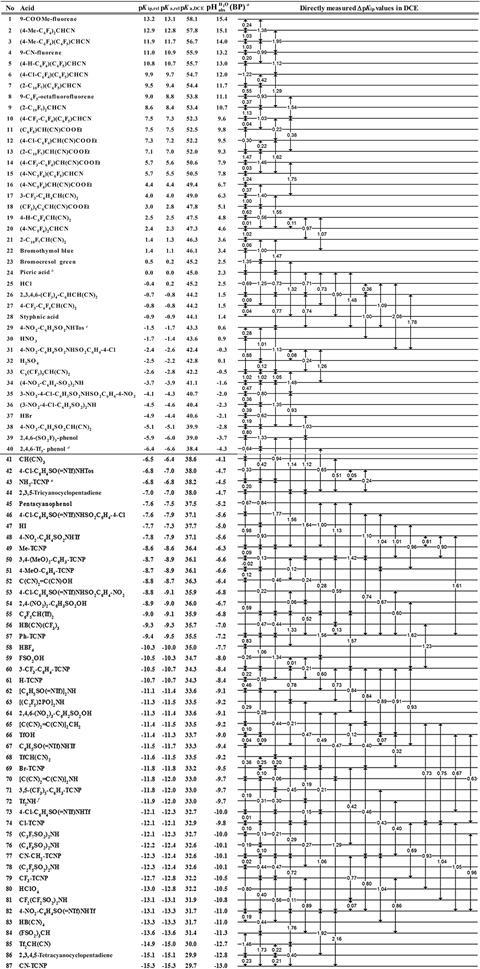
A collaboration between scientists in Estonia and Germany has resulted in a comprehensive solvent acidity scale spanning 28 orders of magnitude, twice as much as the classical pH scale.
Organic chemists use the traditional pH scale to select suitable catalysts for acid-catalysed reactions carried out in water. However, acid-catalysed reactions are commonly carried-out in non-aqueous solvents of varying polarity. For these solvents, there is no general acidity scale, so trial and error is a common catalyst selection method. A comprehensive solvent acidity scale would allow for quantitative catalyst selection and give way to more convenient, reliable and reproducible reactions.
Now, Ivo Leito, from the University of Tartu, Ingo Krossing, from the Albert Ludwig University of Freiburg, and their colleagues have created such a scale. It’s based on the measured molecular acidities of compounds relative to picric acid in dichloroethane. Their thermodynamically-accurate wide-ranging solvent acidity scale spans 28 pH units, and corresponds to the ability to slow down or speed up an acid/base-catalysed reaction by a factor of 1028. To make it, they studied more than 80 buffer solutions that give a very wide acidity range for synthetic chemists. It can be applied to many non-polar solvents making this a very accessible method for both lab- and industrial-scale reactions. The researchers say their work will have direct practical uses for synthetic chemists, who will ‘now have a large range of buffer systems that can allow them to set-up constant acidity levels even in very non-polar solvents.’
Christian Reichardt, an expert in solvent effects at the Philipps University of Marburg, Germany, agrees that this work will have wide reaching implications: ‘The impact of this scale is not a very specific but a more general one for all kinds of acid-catalysed reactions. Having a unified Brönsted acidity scale, valid for the non-polar solvent 1,2-dichloroethane, should now allow catalyst selection using more quantitative aspects.’
Leito and Krossing are now interested in how this work, as well as their previous work on a unified acidity scale , which compares acidities in solution as well as in the gas phase and solid state, will help scientists to understand fundamental systems in life sciences and materials sciences.








No comments yet