Repurposed antioxidant’s mechanism-mending properties make tough alkene alkylation possible
The first alkylation of heavily substituted alkenes has been performed thanks to a repair mechanism that captures and removes pesky side products.
‘We have changed the limits of what we can do with radical chemistry in terms of making carbon–carbon bonds,’ says Philippe Renaud from the University of Bern, Switzerland, who led the team behind the research.
Since the discovery of metal catalysed cross coupling reactions, synthesising aromatic molecules has become routine. But many bioactive compounds and potential drugs contain extended saturated scaffolds – to make these, chemists have to come up with different strategies. Radical additions install functional groups across carbon–carbon double bonds to create complex alkanes.
But while this works well on terminal alkenes, it gets harder the more substituted the double bond is. Non-terminal alkenes can form allylic radicals – ‘lazy’ radicals, explains Renaud, that kill off the chain process. Eventually, the side reaction consumes all active radicals, until none are left to keep the reaction going.
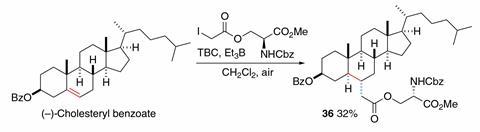
‘We cannot avoid these side reactions, but we can repair them,’ says Renaud. A catechol antioxidant, his team has now found, can kick lazy radicals back into action. This makes it possible to alkylate large, substituted alkenes like cholesterol and even tetramethylethylene – a process that has always been considered ‘extremely difficult’, according to organic chemist Ryan Shenvi from Scripps Research Institute in the US.
‘Efficient intermolecular radical addition to polysubstituted alkenes is a significant achievement,’ says Shenvi, adding that ‘the ease of execution makes [the reaction] very appealing’. Moreover, it could help build complex ring systems. ‘Radical cyclization has proved a useful tactic but can be hard to toggle between different regioisomers. You could imagine reversing the regioselectivity of carbon–carbon bond formation by first carrying out this intermolecular alkene addition and then cyclising at a different site.’
But the finding was not straightforward, says Renaud. In nature, catechols and other antioxidants inhibit, rather than encourage, radical formation. The trick lies in adding the radical initiator triethylborane, which creates highly reactive ethyl radicals as soon as the catechol captures a lone electron during the repair mechanism.
Renaud says his team is now exploring if the reaction works on complex natural products. ‘Other functional groups would stay completely untouched, so we wouldn’t need protecting groups,’ he says. ‘This is the advantage of radical chemistry, which is an extremely mild method.’ Since the reaction prefers electron-rich double bonds, Renaud thinks that they will be able to target one out of several alkene groups within a molecule.
Even other radical chain reactions that suffer from deactivating side reactions ‘may also potentially be repaired by a system of this type’, Renaud says.
References
G Povie et al, Sci. Adv., 2018, DOI: 10.1126/sciadv.aat6031















No comments yet