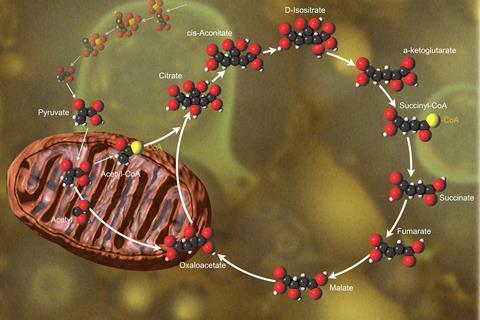
Generations of biochemistry students have learned that the role of the Kreb’s cycle or tricarboxylic acid (TCA) cycle is two-fold: to generate energy for cells and to create the building blocks for growth. But researchers are finding that metabolic pathways – even canonical ones like the TCA cycle – can actually be configured in a variety of ways, and they have many more jobs than previously imagined. Now, researchers have discovered that the TCA cycle has yet another underappreciated role: getting rid of waste.
Depending on cell type and development stage, cells change how they metabolise nutrients. A few years ago, for example, researchers discovered that during infection, immune cells rewire their TCA cycle to make itaconate, an anti-microbial metabolite. Lydia Finley, a cancer biologist at Memorial Sloan Kettering, and her team wanted to know what metabolism is doing in various cells . ‘We know that what you need is different depending on who you are as a cell,’ Finley says. ‘What are the different ways that cells are solving metabolism, and what is metabolism doing for them?’
To examine this question, Finley’s team knocked out one of the enzymes in the TCA cycle, creating a bottleneck that caused the accumulation of citrate, the initiating metabolite in the cycle. Its accumulation signals that there are too many nutrients coming in relative to demand and triggers a stress response, the researchers found. When they knocked out a second enzyme in the TCA cycle – the one that makes citrate – this restored the cells and they grew normally, despite the disruption of energy production through the TCA cycle. The result showed that the ability to prevent citrate accumulation, either by clearing it quickly or by avoiding producing it in the first place, was key to keeping cells healthy.
In mice carrying this TCA cycle mutation, the kidney was the first organ to fail. That’s because the kidney is the only organ that uses citrate as a fuel: it is therefore the one that experiences the biggest problems when citrate can’t be cleared. Surprisingly, the heart and brain, tissues that require a lot of energy, were fine for three weeks, the duration of the experiment, in spite of a broken TCA cycle. This suggested that cells can easily find alternative metabolic routes to produce energy. It was the ability to clear citrate that was the essential job of the TCA cycle. ‘It’s essentially a garbage compactor for the cell,’ says Finley.
‘We tend to think the important thing about a metabolic pathway is the production of the product,’ says University of Utah biochemist Jared Rutter, who wasn’t involved in the research. ‘It’s a surprising phenomenon that, in fact, the most damaging thing you can do with a metabolic pathway is not to block the production of the ultimate product. It’s to actually block it in the middle and accumulate some toxic intermediate. This paper sort of shows mechanistically and with great detail just how true that is, in a way that’s been rarely done before.’
Finley’s findings point to a much broader phenomenon that has clinical implications. Inborn errors of metabolism can lead to rare diseases when patients have germline mutations in metabolic enzymes. Sometimes, disease is caused by the fact that, when an enzyme in a metabolic pathway is defective, the cells cannot make a downstream product. But Finley’s work suggests that patients could often be sick because of the accumulation of a toxic intermediate.
‘This concept is one that is probably not very well appreciated outside of the aficionados of the field,’ Rutter says. Of the findings, he says, ‘the implication goes much beyond citrate and probably there are many examples of this that we don’t yet know about that will be found at some point in the future’.
References
A Xie et al, Cell, 2026, DOI: 10.1016/j.cell.2026.01.028










1 Reader's comment