Click chemistry’s toolbox has a new technique: phosphorus fluoride exchange (PFEx). This reaction enables scientists to easily build oxygen and nitrogen-linked products from a central phosphorus hub.
Inspired by nature’s penchant for phosphate connections, US-based chemists John Moses of Cold Spring Harbor Laboratory in New York and two-time Nobel laureate Barry Sharpless of the Scripps Research Institute in La Jolla, California, replicated their previous innovation of sulfur fluoride exchange (SuFEx) with phosphorus. ‘Just one step to the left and the world is different,’ says Sharpless. ‘It worked better than we imagined,’ adds Moses.
Click chemistry is what won Sharpless his second Nobel prize in chemistry. It’s characterised by using a set of reliable, high yielding and clean reactions, such as the copper-catalysed azide–alkyne cycloaddition, to snap together small modular chemical units. Phosphate linkages are prevalent in biomolecules, serving as the connectors in DNA, RNA and ATP, but existing techniques for synthesising phosphorus linkages typically employ nucleophilic exchange of P–Cl containing electrophiles that are prone to over-substitution and degradation.
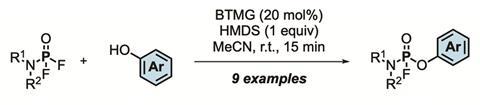
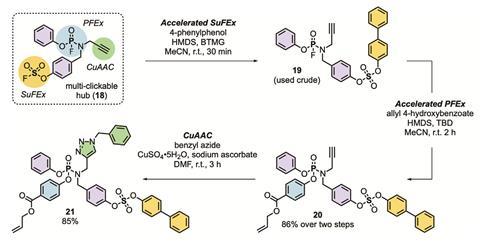
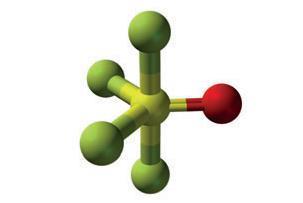
PFEx reactions instead take advantage of the improved reactivity profile afforded by the shorter length and higher stability of P–F bonds over P–Cl bonds. Molecules featuring multiple P–F bonds undergo an efficient Lewis-base-catalysed exchange with incoming aryl alcohols, alkyl alcohols, and amine nucleophiles. In this way, the tetrahedral phosphorous core is selectively, sequentially, and quickly decorated with P–O and P–N linked functional groups. However, the researchers note that the organofluorophosphate compounds required for PFEx contain functional groups that appear in some nerve agents, and anyone attempting to use the technique should follow strict safety protocols.
‘A key concept in click chemistry is the ‘hub’: devising an entry point into the controlled reactivity of an atom or moiety,’ explains Han Zuilhof, an organic chemist based at Wageningen University in the Netherlands. ‘In the case of phosphorus, the use of the P–F bond endows this control … it tames the phosphorus atom like a good jockey [tames] a fast and potentially jumpy horse.’
Importantly, PFEx is done under mild conditions and operates orthogonally to other click reactions, allowing the new technique to function harmoniously alongside the established reactions that Moses and Sharpless call ‘near perfect’.
‘The ability to synthesise diverse P–F compounds, as well as demonstrate their reactivity, has tremendous potential for expanding click chemistry in synthesis,’ says Nicholas Ball, a synthetic chemist from Pomona College in California, US. ‘Following the footsteps of SuFEx, [Sharpless and Moses] have instigated our imagination and curiosity of what we can do with phosphorus compounds.’
The researchers believe the high speed of click chemistry reactions could reduce the time researchers spend on synthesis, enabling them to focus more on screening molecules for toxicity and function. ‘If we do this the click chemistry way, there is no process chemistry,’ says Sharpless. ‘In the world of Kung Fu, speed determines the winner,’ he adds, invoking a line from the film Kung-Fu Hustle.
The researchers hope that greater diversity in clicked molecules will lead to discoveries, including new bioactive molecules. ‘P–F chemistry has a history of bioactivity: [these compounds] were used as nerve agents. That means we know that they have biological function and that should be scary, initially, but in retrospect, we should consider that to be a benefit,’ says Moses. Sharpless is focused not on what molecules PFEx could make but rather the effects those molecules could have: ‘It’s not just a molecule or an enzyme. It’s a function we’re after.’
Update: This article was amended on 20 June 2023 to highlight the safety considerations around working with organophosphorus compounds.
References
S Sun et al, Chem, 2023, DOI: 10.1016/j.chempr.2023.05.013












No comments yet