We take a look back at the year’s most interesting chemical science stories
Which chemistry stories got us talking in 2014? With the help of an expert panel of journal editors Chemistry World looks back at some highlights from last year’s ground breaking chemical sciences research.
The name’s Bond
Over the past year, our pages have seen plenty of work aimed at advancing our understanding of chemical bonds.

A group led by Thomas Albrecht-Schmitt at Florida State University in the US investigated the bonding of californium – the heaviest element that exists in usable quantities on Earth. Using a sample of several milligrams – with a monetary value of more than a million dollars – they showed that californium, previously thought to be dominated by ionic interactions as in most f-block elements, can actually form covalent bonds to borate.1 The resulting compound has unusual properties, arranging into sheets with distorted borates surrounding each californium centre. The work could have implications for the nuclear power industry, especially for separating americium, curium, berkelium and californium in spent reactor fuel.
More recently, the existence of an entirely new type of chemical bond – first proposed in the early 1980s but never confirmed experimentally – was reported by an international team of researchers.2 They used different isotopes of hydrogen to explore the reaction between bromine and hydrogen bromide (HBr) to create the radical BrHBr. When heavier hydrogen isotopes were used, bonding in the triatomic system is through classical van der Waal’s interactions. But when an atom of ultralight muonium is sandwiched between two heavier bromine atoms, the system is stabilised by ‘vibrational bonding’, with the light atom shuttling between its two neighbours.
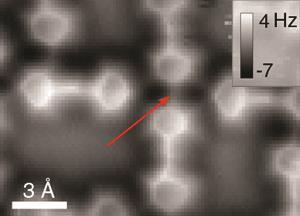
This time last year, we highlighted stunning atomic force microscopy (AFM) pictures that appear to show hydrogen bonds between quinolones as one of 2013’s most exciting stories. But now, controversy has erupted over the interpretation of the images, as other groups’ observations suggest these images of hydrogen bonds may be an artefact. Researchers in Finland and the Netherlands obtained AFM images showing similar features between nitrogen atoms that do not hydrogen bond to each other.3 The team’s findings suggest the narrow high-contrast lines seen between molecules aren’t high electron density areas, but ridges in the ‘interaction potential’ – the force the carbon monoxide AFM tip and the molecules exert on each other. The authors of the original paper have welcomed work towards better understanding of the technique, but are unconvinced by this explanation. With any luck, future studies will help to clarify the situation.
Celebrating crystallography

2014 saw crystallography take the spotlight, having been named the International Year of Crystallography by the United Nations Educational, Scientific and Cultural Organization (UNESCO).
The year was chosen as it is close to the anniversaries of two major landmarks in the field’s history. Just over a hundred years ago, father-and-son team William Henry and William Lawrence Bragg first showed that diffracted x-rays can be used to map the positions of atoms within a crystal and determine its three-dimensional structure. It is also around 400 years since one of the earliest studies of crystal structures: astronomer Johannes Kepler’s observations of ice crystals in snowflakes.
The year-long celebration aimed to both strengthen international collaboration among crystallographers through a series of scientific meetings and to raise the discipline’s profile among the general public with educational activities taking place throughout the year. These included a worldwide crystal growing competition for schools and a crystallography-themed exhibition from photographer Max Alexander at the Royal Albert Hall in London, UK. This year the beacon will be passed to physicists and spectroscopists, for the International Year of Light.
The flat family
Ever since the discovery of graphene, researchers have been searching for two-dimensional equivalents of other elements. In 2014 there was a bumper crop, starting with a group led by Peide Ye at Purdue University in the US, who made phosphorene, a phosphorus analogue of graphene, using the ‘Scotch tape’ method to exfoliate layered black phosphorus in much the same way as graphene can be made by exfoliating graphite.4 They say the material – an electron-poor or p- type semiconductor – could have even better electrical properties than graphene and were able to build transistors with it.

Some months later, the existence of a different form of phosphorene – a two dimensional version of blue phosphorus – was predicted using theoretical calculations by Zhen Zhu and David Tomanek at Michigan State University.5 Blue phosphorene, the team say, would have a similar structure to black phosphorene but the layers would be flatter and bear a closer resemblance to graphene with shallower ‘zig-zag’ ridges as opposed to the deep, trench-like ridges of black phosphorene.
Silicene – the two-dimensional allotrope of silicon with a graphene-like honeycomb structure – was first grown back in 2012, but this year theoretical chemists predicted a variation made of silica (SiO2), silicatene.6 The team calculated that adding oxygen to the silicene lattice should produce a stable monolayer of silica with some interesting mechanical and electrical properties. Silicatene would get thicker when stretched, and would have a large, direct band gap of about 3eV. Stretching the material along one crystalline axis would increase this band gap, while stretching it in the other direction would reduce it.
Further additions to the flat materials family include germanene – a one-atom thick layer of germanium – which was grown by dry deposition of germanium onto a gold substrate by an international team led by Guy Le Lay at Aix-Marseille University in France.7 And, finally, arsenene – a graphene-like arsenic monolayer – whose potential properties have been explored using theoretical calculations by Chinnathambi Kamal from Raja Ramanna Centre for Advanced Technology, India, and Motohiko Ezawa, University of Tokyo, Japan.8
As well as novel 2D allotropes, 2014 saw researchers successfully create 2D crystal polymers for the first time. Two groups, based at ETH Zurich in Switzerland and the University of Nevada in the US, independently showed that monolayers of propeller-like anthracene-based monomers can be fused into flat, one molecule thick polymeric sheets by UV light.9,10 These 2D polymers should have more complex chemistry than graphene and could, eventually, find uses as highly selective molecular sieves or within 2D electronics.
Solar jet fuel

It sounds like wishful thinking – generating jet fuel out of thin air using the power of sunlight. But scientists in Europe came pretty close to doing just that when they made kerosene out of water and carbon dioxide in a concentrated solar reactor.
The group of Aldo Steinfeld at the Swiss university ETH Zurich, together with colleagues in the EU-backed Solar-Jet project, used a high-flux solar simulator, which mimics the output of an actual solar concentrator, to produce the high temperatures needed to split carbon dioxide and water into hydrogen, carbon monoxide and oxygen. The hydrogen and carbon monoxide mixture, known as synthesis gas or syngas, can then be converted into kerosene via the Fischer–Tropsch process. One breakthrough was the team’s use of cerium oxide to harness oxygen, which would otherwise be dangerously explosive, so that it can be separated from the syngas and safely removed.
The technology is still in the early stages and so far the group have only made enough kerosene to fill a glass jar. They will now spend the next few years trying to scale up the process and increase the efficiency enough to make it economically viable. The long term goal is to produce 20,000 litres of kerosene per day in a square-kilometre-sized solar tower system.
Better batteries

Speaking of electronics, we’ve heard about various groups working to improve batteries, hoping to make them more efficient, greener and last longer.
Elton Cairns, and colleagues at the Lawrence Berkeley National Laboratory in California, US, made a lithium-sulfur battery that can hold more than double the energy of ordinary lithium-ion batteries weight-for-weight.11 They overcame the problem of lithium–sulfur batteries losing energy when repeatedly recharged by sandwiching the sulfur electrode between a layer of graphene oxide, to immobilise it, and a protective outer layer of cetyltrimethyl ammonium bromide, which preserves the sulfur by stopping soluble lithium polysulfide intermediates forming in the electrolyte. The resulting batteries continue to function well even after being recharged 1500 times, and the team say they could hold enough power to keep an electric car going for over 300 miles.
Percival Zhang’s group at Virginia Tech in the US has taken a different approach, building a bio-battery that runs on sugar.12 Their prototype can also hold twice as much energy as ordinary lithium ion batteries and could one day be used to power mobile phones or other small devices. The AA-sized prototype is powered using maltodextrin – the researchers created a novel enzyme pathway that strips electrons off the sugar and transfers them to electrodes to generate power. The system can be recharged simply by topping it up with more sugar solution, a safer and more abundant resource than lithium.
Meanwhile, novel materials are giving traditional lithium-ion batteries interesting new properties. Huisheng Peng and colleagues at Fudan University in China developed a battery that can be stretched by 600%.13 To make the electrodes they wound two carbon nanotube–lithium oxide composite yarns onto an elastomer substrate and then covered this with a layer of gel electrolyte. One day, batteries like these could be woven into textiles to help meet the growing demand for wearable electronics. And the groups of Yi Cui and Zhenan Bao at Stanford University in the US have created battery anodes that can heal themselves when they crack, made from silicon microparticles embedded in a self-healing polymer that joins up the margins of any fractures that appear.14
We’ve also seen improvements to battery safety. An early warning system that can help prevent batteries overheating and catching fire was developed by a team based in the US and China.15 The group designed a smart ‘separator’ sheet that can detect lithium dendrites – spongy structures that grow on the anode as the battery is used – and prevent the two electrodes coming into contact and short circuiting. And a team at Harvard University in the US developed a pressure-sensitive coating for button cell batteries that prevents them harming curious children that accidentally swallow them.
Buckyball boron

After several papers predicting the structure, researchers observed thefirst experimental evidence of borospherene, a box-like boron fullerene with the formula B40.17
Unlike carbon, boron can’t form a perfect football like Buckminsterfullerene (C60) because it only has three electrons in its outer shell, so after forming three bonds it has no free electrons remaining to form the delocalised p-network essential to the stability of C60. But various theoretical studies have showed that other configurations may be stable.
Lai-Sheng Wang at Brown University, US, calculated that 40 borons could form a box-like fullerene isomer with both hexagonal and heptagonal holes. Their simulations suggested that gaseous B40 would exist as a mixture of two forms: a quasiplanar structure and the fullerene. The team vaporised bulk boron with a laser and separated the molecular clusters using a mass spectrometer. They calculated the predicted photoelectron spectra of both quasiplanar and the fullerene B40- isomers and compared these with their measured spectrum. The measured spectrum did not match either calculated spectrum, but matched a combination of the two. This indicates, consistent with their predictions, a mixture of the quasiplanar isomer and borospherene.
Life’s beginnings
As ever, chemists continue to ponder the big questions surrounding the origins of life.
Did life begin in a tiny water droplet? That was the question Sylvain Ladame of Imperial College London, UK, and colleagues investigated by comparing reaction rates between small organics within droplets, compared with free standing water. The thinking is that, on the early Earth, small molecules would have been brought much closer together within droplets and stood a greater chance of reacting together to form more complex ones, eventually leading to life. Ladame’s group showed that the reaction rate between two small molecules was 45 times faster when they were enclosed in tiny microdroplets, compared with freestanding water, suggesting these environments may have offered ideal conditions to support the emergence of life.18

Another group has suggested that the kind of metabolic pathways found within living organisms began in ancient oceans around 4 billion years ago, and were catalysed by metal ions, not enzymes. Markus Ralser and colleagues at the University of Cambridge, UK, looked at simple reactions found in sugar phosphate metabolism under different early Earth scenarios. They found 29 reactions could be catalysed by metal ions, including ferrous iron which appeared to be the most effective catalyst.19 As iron is thought to have been abundant in early oceans, their work offers a plausible mechanism for how these pathways might have originated and subsequently been incorporated into living cells.
And some new information about the ancient world has been revealed by looking at ancient samples. US scientist Stanley Miller’s simulation experiments in the 1950s showed how amino acids could have arisen from the primordial soup. But some of his samples were forgotten and never analysed. Now, Miller’s former doctoral student Jeffrey Bada has revisited some of these samples after more than 50 years and found they provide a possible explanation for how the first amino acids polymerised into the first proteins. Miller had repeated his original experiment adding cyanamide, a condensing reagent used to produce pharmaceuticals. When Bada’s group analysed the resulting samples they found 10 dipeptides had been produced. Although cyanamide usually only polymerises amino acids under acidic conditions, the group found that some of the intermediates produced in the experiment would do so under the alkaline conditions of early Earth.20 The resurrection of these much earlier experiments may further our understanding of how the first building blocks of life emerged millions of years ago.
Life’s alphabet extended
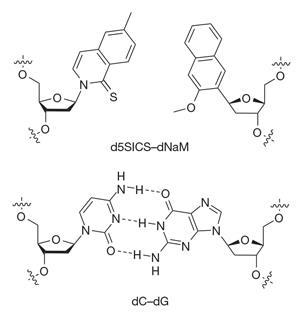
Every science student is taught that within living organisms, DNA is made up of two base pairs – adenine and thymine (A–T) and cytosine and guanine (C–G). But Floyd Romesberg and colleagues at the Scripps Research Institute in the US have rewritten this rule by engineering a ‘semi-synthetic’ strain of Escherichia coli that contains an additional base pair, which they dubbed X–Y.21 Their bacterial strain is the first organism that can grow and replicate with an unnatural base pair in its DNA.
The group had been trying to engineer artificial nucleotides for decades – the X and Y bases are two molecules called d5SICS and dNAM that pair up using hydrophobic interactions. The team were able to synthesise a bacterial plasmid containing a single X–Y base pair, which was successfully taken up by E. coli cells. The cells could then replicate when ‘fed’ the new base pairs in the growth medium. The team say their breakthrough opens up the possibility of creating new proteins or finding new ways to regulate genes within cells. Now that this milestone has been achieved, it will be exciting to see what synthetic biologists are able to do with it.
Crisis chemistry
From the ancient earth to today’s news, chemistry has played a crucial role in events that have dominated headlines around the world this year.

Although civil war continues in Syria, the majority of its declared stockpile of chemical weapons has been destroyed following the government’s decision to join the Chemical Weapon Convention in response to international pressure. Over 1000 tonnes of chemical weapons – including sulfur mustard and precursors for the nerve agents sarin, VX and VM – have been chemically neutralised or incinerated in a massive operation that involved teams from several countries. Almost a year after a sarin attack killed around 1500 civilians in a Damascus suburb, it was announced that disposal operations in the UK, Finland and on board the US Navy ship Cape Ray had been completed.
And in early 2014, news of a serious outbreak of the Ebola virus in West Africa began to reach us. This has since escalated into a large-scale epidemic, to which scientists and health professionals have been struggling to respond. The World Health Organization has sanctioned the use of experimental drugs such as antibody treatment ZMapp to fight the disease, and programmes to develop a vaccine have been fast tracked in several countries. The US government will provide technical expertise and millions in funds to Maryland-based Profectus BioSciences to speed up tests on a potential Ebola vaccine. And pharma giant GlaxoSmithKline, in concert with the US National Institutes of Health, has completed Phase I trials of another vaccine, and plans to start the next phase of trials in 2015. Let’s hope progress continues into the coming year.
And with that we bring an end to our roundup – as always, a mere snapshot of last year’s research. We look forward to seeing more in 2015.
Correction: On 12 December an incorrect reference to boron in the vibrational bonding story was replaced with bromine
References
- M J Polinski et al, Nat. Chem., 2014, 6, 387 (DOI: 10.1038/nchem.1896)
- D G Fleming et al, Angew. Chem., Int. Ed., 2014, 53, 13706 (DOI: 10.1002/anie.201408211)
- S Hämäläinen et al, Phys. Rev. Lett., 2014, 113, 186102 (DOI: 10.1103/physrevlett.113.186102)
- H Liu et al, ACS Nano, 2014, 8, 4033 (DOI: 10.1021/nn501226z)
- Z Zhu and D Tomanek, 2014, arXiv:1403.1300v3
- V Ongun Özçelik, S Cahangirov and S Ciraci, Phys. Rev. Lett., 2014, 112, 246803 (DOI: 10.1103/PhysRevLett.112.246803)
- M E Dávila et al, New J. Phys., 2014, 16, 095002 (DOI: 10.1088/1367-2630/16/9/095002)
- C Kamal and M Ezawa, arXiv:1410.5166v1
- M J Kory et al, Nat. Chem., 2014, DOI: 10.1038/nchem.2007
- P Kissel et al, Nat. Chem., 2014, 6, 779 (DOI: 10.1038/nchem.2008)
- M-K Song, Y Zhang and E J Cairns, Nano Lett., 2013, 13, 5891 (DOI: 10.1021/nl402793z)
- Z Zhu et al, Nat. Commun., 2014, 5, 3026 (DOI:10.1038/ncomms4026)
- Y Zhang et al, J. Mater. Chem. A, 2014, 2, 11054 (DOI: 10.1039/c4ta01878h)
- C Wang et al, Nat. Chem., 2013, 5, 1042 (DOI: 10.1038/nchem.1802)
- H Wu et al, Nat. Commun., 2014, 5, 5193 (DOI: 10.1038/ncomms6193)
- B Laulicht et al, Proc. Natl. Acad. Sci. USA, 2014, 111, 16490( DOI: 10.1073/pnas.1418423111)
- H-J Zhai et al, Nat. Chem., 2014, 6, 727 (DOI: 10.1038/nchem.1999)
- A Fallah-Araghi et al, Phys. Rev. Lett., 2014, 112, 028301 (DOI: 10.1103/physrevlett.112.028301
- M A Keller, A V Turchyn and M Ralser, Mol. Syst. Biol., 2014, 10, 725 (DOI: 10.1002/msb.20145228)
- E T Parker et al, Angew. Chem., Int. Ed., 2014, 53, 8132 (DOI: 10.1002/anie.201403683)
- D A Malyshev et al, Nature, 2014, 509, 385 (DOI: 10.1038/nature13314)












No comments yet