Twisted structure is an unprecedented case of a compound that becomes aromatic by breaking its own symmetry
Chemists have created the first-ever half-Möbius molecule. The orbitals of the 13-carbon ring twist by only a quarter turn with each circumnavigation rather than the half-twist of regular Möbius aromatics. It’s also the first example of a molecule that becomes more aromatic as it becomes less symmetric.
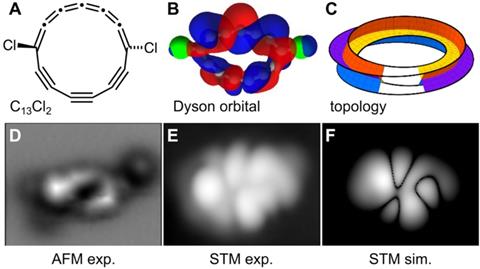
A team led by Igor Rončević from the University of Manchester, UK, and Leo Gross from IBM in Zurich, Switzerland, first spotted the curious structure – C13 Cl2 – in 2024, as an intermediate in their synthesis of cyclo[13]carbon. They were zapping individual molecules of a tricyclic, fully chlorinated precursor with the tip of a scanning tunnelling microscope (STM) to remove the chlorine atoms.

‘We noticed [this intermediate] was chiral – that was the first excitement,’ recalls Harry Anderson from the University of Oxford, UK. ‘You can apply voltage pulses to switch it between enantiomers. That fascinated us and we realised that the orbitals have this strange half-Möbius topology.’
The aromatic π-orbitals in Möbius molecules have a 180º twist for every loop around the ring while the orbitals in the new half-Möbius molecule twist only by 90º – meaning they need to wind around the ring four times to return to the start. This is because they have a +-shaped cross-section, unlike the linear orbital cross-section in a regular Möbius ring.
‘It’s not often that you get to read a paper that introduces truly new concepts and this is very much one of them,’ comments Henry Rzepa from Imperial College London, UK, an expert on Möbius aromatics who wasn’t involved in the project. ‘[It’s] a remarkable achievement across a number of dimensions: organic chemistry, surface science, nanoscience and quantum chemistry,’ adds theoretical chemistry expert Gemma Solomon from Denmark’s University of Copenhagen.
The C13 Cl2 molecule also has a planar, weakly anti-aromatic form that is less stable than the two buckled, half-Möbius enantiomers. ‘As it breaks its symmetry, it becomes aromatic,’ Rončević explains. ‘This has never been observed to my knowledge. Usually, as we distort something it becomes less aromatic or less anti-aromatic.’
‘It’s a horribly complicated problem in terms of electronic structure,’ Rončević adds. ‘The workhorse of modern computational chemistry, DFT, is completely unreliable for this molecule.’ The team decided to run more accurate calculations on IBM’s quantum computer in what Solomon calls ‘one of the most understated applications of quantum computing hardware’.
Currently, each half-Möbius ring is ‘hand-crafted’ with an STM, on a salt surface, in ultra-high vacuum, at –268ºC. Encapsulating the molecules in macrocycles might make them more handleable, but Gross is sceptical whether they hold real-world applications. He’s more interested in exploring other curious structures: ‘What are the principles to stabilise them? Can you go even further, having multiple twists or braided twists?’
Not every new compound needs to have an immediate use, adds Rzepa. ‘One of the real-world applications is that it’s giving people new ideas.’
References
I Rončević et al, Science, 2026, DOI: 10.1126/science.aea3321














No comments yet