A new drug delivery technique uses synthetic molecular motors to release a drug when activated by visible light. The proof-of-principle delivery mechanism, which was tested on cultured lung cancer cells, could enable a non-invasive, highly controlled and targeted way to activate drugs inside the body.
Ben Feringa at the University of Groningen in the Netherlands is credited with making the first light-powered synthetic molecular motor in 1999. His subsequent work, including a molecular nanocar, earned him a share of the 2016 Nobel prize in chemistry. Other groups have used motors based on Feringa’s work to explore therapeutic applications, including drilling holes into cancer cell membranes to kill them.
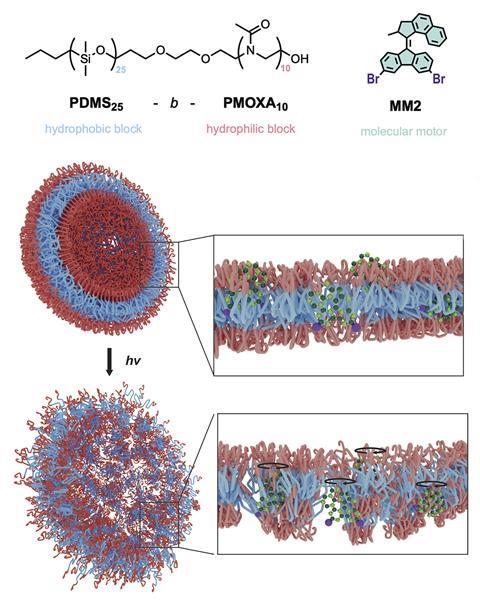
Now, Feringa’s lab has teamed up with Cornelia Palivan’s group at the University of Basel, Switzerland, to develop a drug delivery technique that incorporates molecular motors into cargo-carrying artificial vesicles called polymersomes, which are made from diblock copolymers and resemble the phospholipid bilayer membrane of living cells. The concept is simple: the vesicles are loaded with a drug cargo and, when activated by light, the molecular motors start to spin creating holes in the vesicle membrane to release the drug.
‘The most challenging aspect was to find a suitable block copolymer,’ says Feringa. ‘It had to form a thin bilayer and not be too rigid otherwise the motor would be ineffective.’ He explains that the copolymers also couldn’t be too flexible because the membrane would simply reorganise itself before the drug cargo could be released.
The team focused its attention towards bioinspired polymersomes developed by Palivan’s lab, comprising hydrophobic poly(dimethylsiloxane) and hydrophilic poly(2-methyl-2- oxazoline) (PDMA and PMOXA, respectively). These have previously shown promise for several biological applications and so the team set out to embed light-responsive molecular motors into the artificial vesicle membranes. As the motors are hydrophobic, they could successfully be incorporated into the hydrophobic part of the membrane.
‘We were very excited to see the system working just with a flash of light. This was after several attempts of various designs,’ says Feringa. Experiments showed that low-power visible light triggered the delivery of a fluorescent dye with efficiencies up to 75 per cent and the motors could be turned off and on repeatedly over three cycles. Furthermore, only low amounts of motors in the membrane (1 mol%) were needed to promote release.
The researchers went on to test the system on lung cancer cells in culture by loading the polymersomes with an approved antitumour drug. The results showed similar drug action on the cells as other experiments that saw cells receive direct doses of drug. The team also established that the polymer and the molecular motors were non-toxic at the concentrations used and that the presence of the molecular motors and light are necessary to promote drug release.
‘The system proposed in this paper exhibits some particularly appealing properties,’ comments Alberta Ferrarini, an expert on membrane dynamics from the University of Padua, Italy. However, she believes more work is needed to fully understand the action of the drug delivery system. ‘The underlying mechanism is not clear and in my opinion a major question, for both application and fundamental science, is the identification of the role of distinct features of the molecular motor, especially of its unidirectional rotation.’
‘Whilst there is still work to do to extend this to the near-IR to enable significant tissue penetration, whether by two-photon techniques or motor modifications, this is clearly a step towards using molecular motors in the clinic - a very exciting goal,’ comments Charlie McTernan who investigates biomedical applications of molecular machines at Kings College, London, UK. ‘It will be interesting to see how the pharmacokinetics of different molecular motors affects future applications; and to see how continuous rotation brings advantages as compared to the use of simple switches or photocleavable units.’
References
A Guinart et al, Proc. Nat. Acad. Sci, 2023 DOI: 10.1073/pnas.2301279120









No comments yet