Meera Senthilingam
This week, a compound made from an element that was thought to enjoy being solo. Brian Clegg explains more.
Brian Clegg
Think of helium, and once you get past balloons and squeaky voices, the most obvious characteristic of the element is that this is a ‘noble gas’ – it is almost completely inert. The electrons around atoms come in ‘shells’ – it is the part-empty outer shell that makes reactions likely. With its full shell of two electrons, helium is armour plated against reacting. And yet we are going to consider a helium compound, a molecule that could have a very ancient pedigree, helium hydride.

Given the secret of helium’s nobility, that filled outer shell, it should be no surprise that helium hydride is only stable as an ion, meaning that there are still only two electrons involved, but one is shared in a covalent bond between helium and the proton that is a hydrogen nucleus. The neutral molecule can briefly exist, but only as an excimer, where the excess electron has enough energy to keep it at a higher level.
Although not the kind of substance to be found on the lab shelf, helium hydride is an acid. In fact the proton affinity of the base – the energy given out when helium reacts with a proton – is the lowest known, technically making helium hydride the strongest acid in existence. This is arguably an obscure substance with no useful application, so it may seem strange that helium hydride has anything more than a passing mention in the chemistry textbooks. However, it could well be that helium hydride was the first compound ever to exist.
For the first couple of minutes after the Big Bang, 13.7 billion years ago, there was no matter as we now know it.

What began as an inferno, so energetic that no particles could stay in existence, cooled and expanded to first form a ‘soup’ of quarks and their exchange particles, gluons, and finally became sufficiently cool for ions to form. Once hydrogen, the most common element, had formed, deuterium came soon after it. This in turn made it possible for the more stable helium 4 to form from the fusion of two deuterium ions. There was also a little lithium, but for our purposes, the important aspect is that just minutes into the existence of the universe there were hydrogen and helium ions present, in a rough ratio of twelve to one.
Any change in the behaviour of helium hydride in the early years of the universe could mean tearing up our present models of how stars and galaxies formed
The whole process of formation is thought to have taken place between the three minute and twenty minute marks in the history of the universe, though things would not cool enough for neutral atoms to appear for another 100,000 years. The actual formation of atoms would not be completed until around 370,000 years had elapsed. Until that point the nascent universe would have been an opaque soup of ions, rather than the transparent universe we know today.
Since the mid-1980s it has been thought that helium would have been the first neutral atom to form, and also the first to produce a molecular bond. Reactions probably began when a helium atom joined with a helium ion to form He2+, but this would have been highly unstable, with the many energetic photons around it rapidly reversing the reaction. We now know that the first stable compound to emerge would have been the helium hydride ion. It would have then have gone on to influence the formation of early stars.
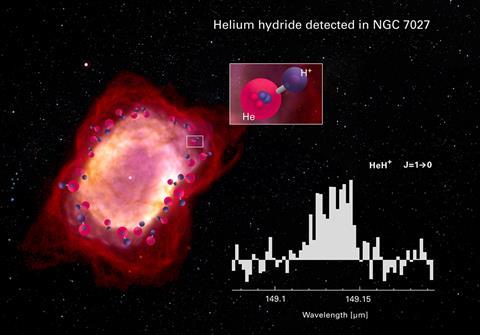
A quandary for astronomers is the lack of definitive evidence. It is thought that examples of the ion should still be out there in space, but the HeH+ is hard to spot with a spectroscope as one of its prominent lines, the ‘fingerprints’ used to identify chemical elements optically, overlaps with lines in the spectrum of the CH methylidene radical. This is frustrating, as models of the development of the universe predict that there should be plenty of helium hydride, both the gaseous nebulae where stars form and in planetary nebulae, clouds of matter thrown off when some types of star are dying. Other molecules predicted by the models have been clearly detected, but not helium hydride (though there is questioned evidence from the nebula NGC 7027).
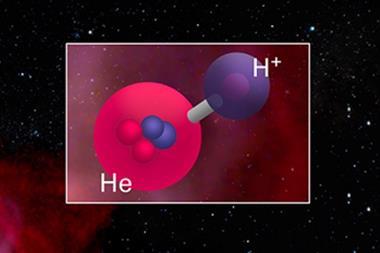
To explain this discrepancy it has been suggested that helium hydride is easier to destroy than the existing models suggest, and specifically that it is more susceptible to being broken down into its components by photodissociation, where an incoming energetic photon breaks the covalent bond holding the structure together.
One suggestion for the cause of this easy photodissociation is that the helium hydride ion can exist in a metastable state, where energy from a photon promotes an electron to a higher level, for longer than was thought. If this is the case it may be more susceptible to taking a second hit while in this metastable state and thus breaking apart more easily than expected. It is hoped that data from the Herschel Space Telescope, which scanned in the infra-red, will cast some light on this mystery. This is crucial, because any change in the behaviour of helium hydride in the early years of the universe could mean tearing up our present models of how stars and galaxies formed.
Helium hydride may have no practical use here on Earth. But this most ancient of molecules is providing a fascinating insight into the way that the universe first evolved, and as such it is a compound with a compelling history.
Meera Senthilingam
Science writer Brian Clegg there, with the chemistry of helium hydride. Next week, show some respect for this compound.
Stephen Wallace
This is more toxic than the famously deadly curare and tetrodotoxin that are found elsewhere in nature, and is over 1000 times more poisonous than cyanide.
Meera Senthilingam
And discover how it can be so lethal by joining Stephen Wallace in next week’s Chemistry in its Element. Until then, thank you for listening, I’m Meera Senthilingam.











No comments yet