Sulfite reductase mimic represents major advancement in engineering synthetic enzymes
Chemists have designed for the first time a haem-[4Fe–4S] catalyst that has similar activity to native sulfite reductase,1 a venture previously hampered by the enzyme’s structural and functional complexity.
Sulfite reductase (SiR) catalyses the six-electron reduction of sulfite to hydrogen sulfide and water – a key part of sulfur metabolism in plants and bacteria – but it’s the ability of sulfide to break down polluting oxyanions such as perchlorate, arsenate and nitrate that makes this enzyme of interest.
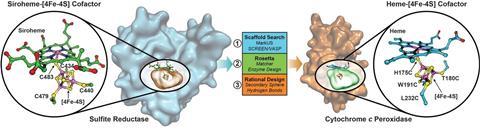
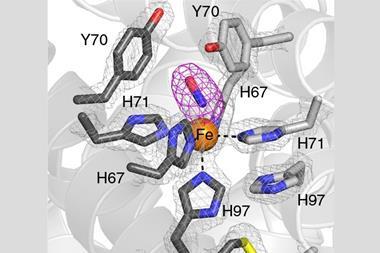
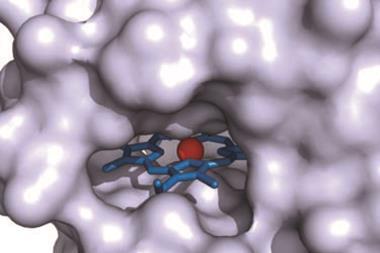
In nature, the cofactor in SiR is unique: SiR acts as a hemoprotein scaffold for [4Fe–4S] clusters that bind covalently to the sirohaem through a bridging cysteine ligand, with lysine and arginine residues surrounding the binding cavity to help protonate sulfide. However, it’s notoriously difficult to design synthetic enzymes for reducing sulfite as the process involves multiple redox reactions and a complex active site with weak sulfide binding.
Now, Yi Lu and his team from the University of Illinois, US, have successfully designed a functional model of SiR using cytochrome c peroxidase (CcP) as a scaffold for binding [4Fe–4S]. CcP is a small stable protein that natively binds haem-b, complete with a cavity that is able to fit the cluster – ideal traits for recreating the catalytic centre of SiR in the artificial enzyme, named SiRCcP. Better still, they were able to tweak the reduction activity of SiRCcP to approach that of the native enzyme, something Lu acknowledges was ‘a challenge’.
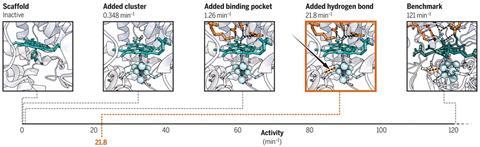
‘We did it in two parts,’ he explains. ‘By introducing secondary sphere interactions – weak non-covalent bonds – we were able to subtly tune the properties of the metal centre. We also introduced positively-charged lysine and arginine residues to stabilise and increase the affinity for sulfite. It’s the combination of the two that made a huge difference in activity’.
The functionality of SiRCcP is promising for using synthetic catalysts to perform multielectron reactions such as sulfite reduction.2 ‘That’s why we’re really excited about the result,’ adds Lu. ‘We can now make an active enzyme with a very complex centre that’s similar to the native version’.
Erin Dodd, an inorganic chemist from the University of Oxford, UK, agrees that is it a ‘very cool’ result. ‘It’s a massive step forward in terms of creating an enzyme almost from scratch that does in fact work… It’s a very important piece of work.’ However, SiRCcP activity may have been even better had native sirohaem been used as a ligand instead of haem-b. ‘I think [the team’s] next logical step would be to try to repeat what they’re doing right now but with the appropriate haem family cofactor,’ she says. ‘This would change the architecture so they might have to do a redesign to optimise the enzyme again’.
References
1 E Mirts et al, Science, 2018, 361, 1098 (DOI: 10.1126/science.aat8474)
2 K Lancaster, Science, 2018, 361, 1071 (DOI: 10.1126/science.aau7754)







No comments yet