Carboxylic acids and esters hydrogenated without the need for expensive precious metals
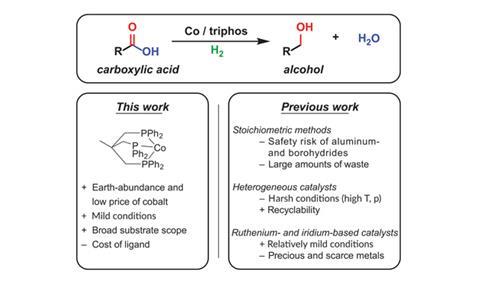
Researchers in the Netherlands have developed a cobalt catalyst that can be used in the hydrogenation of esters and carboxylic acids to alcohols without the need for extremely high temperatures or expensive precious metals.
Bas de Bruin and colleagues at the University of Amsterdam tried out different catalysts made by combining commercially available cobalt salts with a tridentate phosphine ligand in the hydrogenation of methyl benzoate to benzyl alcohol and methanol. The best performing system, which produced high yields under relatively mild conditions, used cobalt tetrafluoroborate hexahydrate (Co(BF4)2·6H2O) as the cobalt precursor. Currently, carboxylic acid hydrogenations – which are used in the synthesis of a variety of desirable compounds including materials and pharmaceuticals – have to be carried out using heterogeneous catalysts at high temperatures and pressures, or far more costly precious metal catalysts based on iridium or ruthenium.
The group say the development, which follows other recent advances in the development of iron and cobalt catalysts for hydrogenation of alkenes, imines and ketones, may enable the production of more high-value chemicals from biomass, as well as making various industrial syntheses more cost-effective.












No comments yet