Scientists in the US have engineered proteins that can catalyse carbon–silicon bond forming reactions – processes never before seen in nature. Such an enzyme could one day offer researchers a way to make silicon-based hybrid organisms typically reserved for the realms of science-fiction.
Despite silicon sitting directly below carbon in the periodic table and sharing many of its chemical properties, no biological organism has ever been known to form a carbon–silicon bond.
Now, a team of scientists led by Frances Arnold of the California Institute of Technology (CalTech), has evolved new enzymes that not only produce organosilicon compounds, but do so more efficiently than the best known synthetic catalysts. The enzymes catalyse the insertion of carbenes into silicon–hydrogen bonds with almost perfect enantiocontrol.
‘This work is about using biology to make carbon–silicon bonds, which is something nature has never done before – even though silicon is the second-most abundant element on our planet,’ says Jennifer Kan, who worked on the project.
The team repurposed cytochrome c ‘heme proteins’, isolated from a bacterium found in Iceland, to catalyse reactions distinct from their natural biological functions as electron transfer agents. ‘We found that when we provide some iron-containing proteins the appropriate sources of carbon and silicon, many of these proteins in fact have the ability to form carbon–silicon bonds,’ explains Kan. ‘The initial activity is often very, very low. But we can significantly improve this ability by doing evolution in the laboratory.’
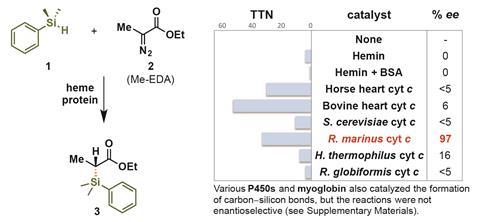
The directed evolution that Kan refers to involved the targeted mutation of particular amino acid groups within the cyctochrome c protein to produce new enzymes with enhanced activities. The team demonstrated that the enzymes could even perform the reactions in vivo, when expressed in E. coli cells.
‘We show that a chemical bond that is not observed in biology can in fact be forged biocatalytically with outstanding efficiency and explicit and tuneable selectivity,’ says Kan. ‘By doing so, we provide biology access to a chemical space that was previously only open to synthetic chemistry.’
Manfred Reetz, a biocatalysis expert from the Max Planck Institute for Coal Research in Mulheim, Germany, says that while many natural and evolved metalloenzymes have been tested in reactions usually associated with synthetic transition metal complexes, examples of enzymes providing both higher activity and selectivity are rare. ‘The advantage of the Arnold system concerns the unusually high turnovers that occur in the protein binding pocket, in addition to fairly wide substrate acceptance,’ he observes.
Kan points out that using renewable and biodegradable enzymes instead of precious metal catalysts also offers environmental benefits. ‘All the chemistry is done under physiological conditions – so in water, under room temperature and neutral pH,’ she says. Kan also notes the surprising stability of the new biocatalysts: ‘Because our enzyme was originally from a bacterium discovered in a hot spring in Iceland, it is super stable – the enzyme can be boiled or autoclaved and still function and perform selective catalysis afterwards.’
Paul Dalby, a biochemical engineer at University College London, UK, believes this enzymatic approach ‘paves the way for significantly more efficient synthesis’ of silicon-containing pharmaceutical compounds, without the need for extensive protecting group strategies.
Speaking in a CalTech video about their research, Arnold ponders the questions that these new biocatalysts could help to answer: ‘We can start asking for the first time: “What happens if you put silicon in place of carbon in living systems?” It’s very hard to explore that chemically unless you have organisms that can make these bonds.’
She is not the only one excited about the possibilities for future biological systems. ‘This work also opens new avenues for synthetic biology in which metabolic pathways could be engineered in for example E. coli, that incorporate silicon atoms selectively,’ muses Dalby. ‘Who knows? We could be looking at the first steps towards synthetic living systems that are carbon–silicon hybrids.’
References
S B J Kan et al, Science, 2016, DOI: 10.1126/science.aah6219










![Designed heme-[4Fe-4S] metalloenzyme](https://d2cbg94ubxgsnp.cloudfront.net/Pictures/380x253/8/8/5/138885_heme_index_2.jpg)














No comments yet