Large and flexible polycyclic ring systems can effectively turn themselves inside out, giving rise to an overlooked phenomenon termed ‘homeomorphic isomerisation’. This conformational process enables structures to switch between distinct forms and could have applications in metal separation and drug delivery, as well as significant implications for intellectual property.
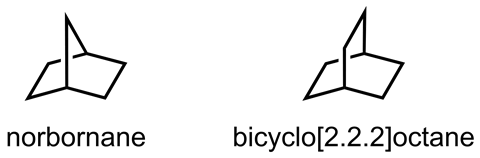
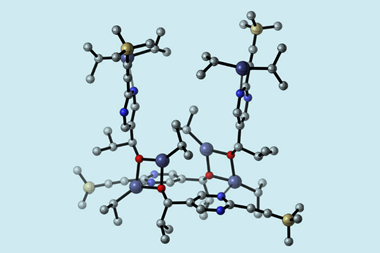
The orientation of the bridgehead atoms in fused ring systems plays a crucial part in determining the molecules’ overall spatial arrangement. In smaller bridged bicycles, the rigidity of the tethering chains often results in conformational locking, where constrained structures like norbornane or bicyclo[2.2.2]octane become fixed into a single orientation. However, the flexibility of larger structures permits a greater degree of freedom, to the point that some are able to invert completely at their bridgehead atoms. ‘Basically, we need two bridgehead entities and three tethers, and we usually need to have at least 10 atoms in these tethers. That’s the minimum requirement,’ explains John Gladysz an organometallic chemist at Texas A&M University in the US.
With the appropriate activation, one tether passes through the ring created by the other two, inverting the entire ring system by rotating the orientation of the bridgehead groups from in to out, or vice versa. How rapidly this process occurs varies between ring systems, and this distinction offers the potential for various applications, says Gladysz.
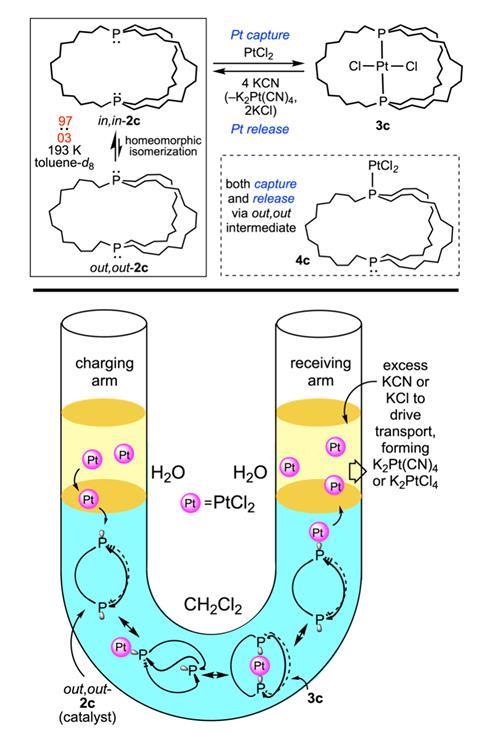
As an example, Gladysz’s team predicted that the phenomenon could aid the separation of metal ions. They filled the bend of a U-tube apparatus with an organic layer containing a rapidly interconverting diphosphine macrobicycle, topping off each arm with an aqueous solution of either platinum chloride or potassium cyanide. Within several hours, the diphosphine ring system had sequestered the bulk of the platinum from one arm to the other, leaving other contaminating metal ions untouched. ‘The out–out form picks up the platinum from the interface, tucks it inside [as the in–in form] for transport,’ explains Gladysz. The reverse homeomorphic process then delivers the platinum at the other end, where it’s trapped by the cyanide.
Drug delivery
However, of most interest to Gladysz is the potential to harness slow interconversion for drug delivery. Macrobicyclic peptides are an increasingly popular motif in drug discovery programmes, but their structures are typically vulnerable to digestive enzymes, making them unsuitable for oral delivery. According to Gladysz, a robust homeomorph of a particular peptide could theoretically be designed to survive the conditions of the digestive tract and pass through the epithelium. This would then convert over a period of hours into an alternative active form that exhibits the required biological effect.
For Andrei Yudin, a synthetic chemist at the University of Toronto in Canada, this is an interesting conceptual approach but a number of challenges remain before it becomes a viable strategy. ‘You want to have this chameleonic property where it changes between states, but how many differentiated conformational states can be controlled?’ he asks. ‘Additionally, sometimes a very small amount of a given conformation is actually responsible for the biological activity, so how do you pinpoint the conformation that is responsible for a particular property?’
Of more immediate impact are the implications of this conformational effect for intellectual property, particularly where a high barrier to interconversion effectively creates two entirely distinct ring topologies. Given the extensive coverage of enantiomerism and polymorphism in patent protection, Gladysz was astonished to find no mention of homeomorphic isomerisation in any of the documents the team evaluated, and believes this could represent a vulnerability in existing IP.

However, while Yudin agrees conformation can have a powerful impact on a compound’s properties, he disputes whether homeomorphism could be legitimate grounds for a claim. ‘If you have composition of matter coverage in a patent, I personally feel that those claims entitle you to assume that you are also covering all possible conformations, because conformation is structure,’ he says.
Regardless of the legal outcome, Gladysz hopes the discussion will bring this overlooked phenomenon back into focus and encourage researchers in all areas to search for and exploit these isomers in their respective fields.
References
I J Smith, S M Popovic and J A Gladysz, J. Am. Chem. Soc., 2026, DOI: 10.1021/jacs.5c21032













No comments yet