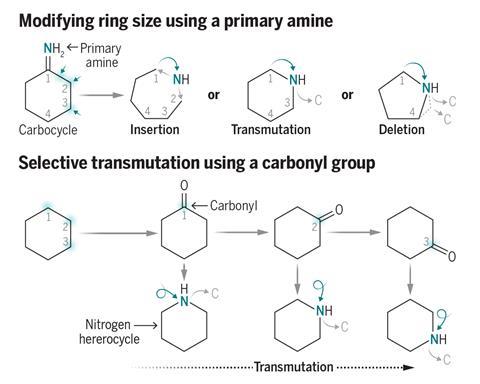
Two new skeletal editing techniques offer a way to create an array of nitrogen-containing rings. Nitrogen heterocycles are valuable structures in many drug molecules, with the researchers behind the studies saying that these synthetic strategies could help accelerate the discovery of candidate drug compounds.
More than 80% of new small molecule drugs approved by the US Food and Drug Administration between 2013 and 2023 contained nitrogen heterocycles, such as piperidines. Chemists often make such rings through the Beckmann rearrangement, which involves reacting a cyclic oxime with an acid to create an amide. However, this approach suffers from poor regioselectivity, requires harsh reaction conditions to reduce the amide to the amine and only leads to ring expansion.
Equally, ‘indoles are highly important structural motifs in pharmaceuticals and natural products’, says Huiying Zeng at Lanzhou University in China. Adding substituents onto the pyrrole ring of an indole can alter the biological and physical properties of such compounds, but the ‘aromatic stability [of indoles] makes precise modification of the core skeleton challenging’, says Zeng.
Zeng’s team has now developed a method that intramolecularly transfers substituents to the C2 position of indole rings.1 Equally, another team led by Lumin Zhang at the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences is using an iodine-based catalyst to make nitrogen-containing rings of various sizes and types from primary amines.2
‘These two [studies] are a wonderful demonstration of the creativity and innovation that the concept of skeletal editing is inspiring,’ says Richmond Sarpong at the University of California, Berkeley in the US, who was not involved with either study.
Adaptable amines
Skeletal editing techniques often struggle with aliphatic compounds and are only capable of a single transformation, says Zhang. He explains that their new method is instead able to produce a range of nitrogen-containing heterocycles from primary amines through a common imino ether intermediate. ‘From the same starting material, we can achieve [products] B, C, D and so on,’ Zhang says. He hopes that this will speed up drug discovery by making it easier to introduce these rings into molecules and study how they affect the properties of drug compounds. ‘I want to push the boundary of skeletal editing,’ he adds.
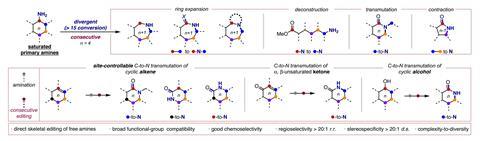
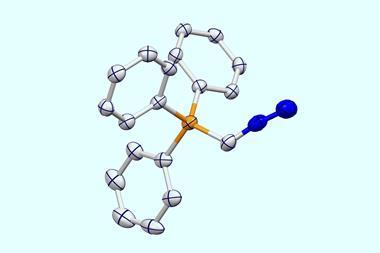
Key to the team’s reaction is a hypervalent iodine catalyst, which has a central iodine atom with three substituents. Zhang says that this catalyst is a mild reagent and can be easily synthesised; the team has so far produced the iodine compound on a 100g scale.
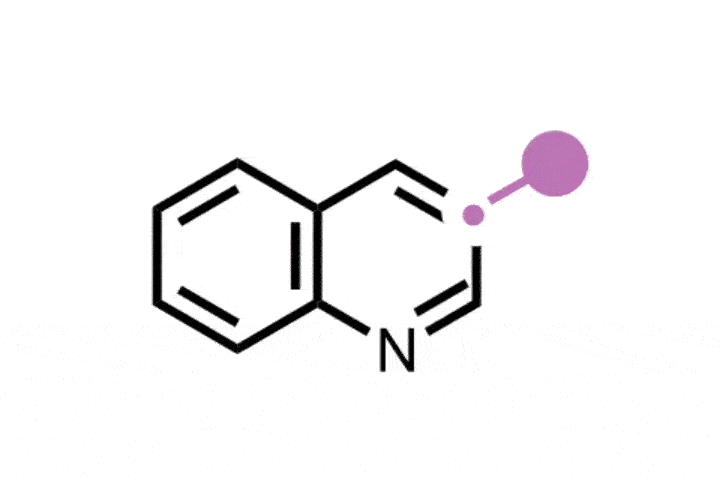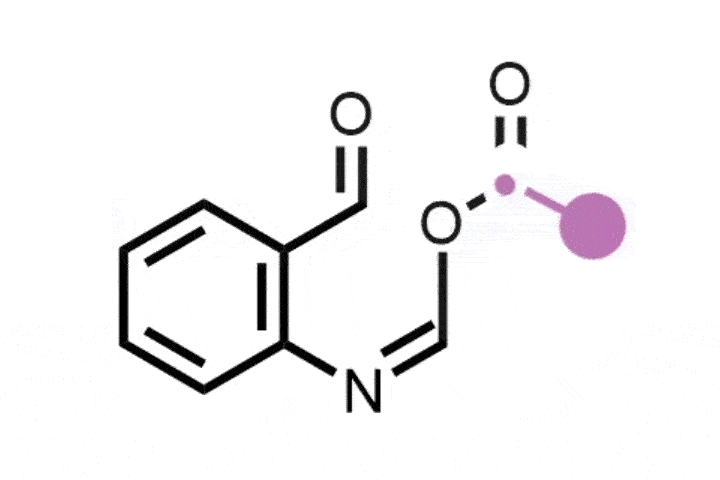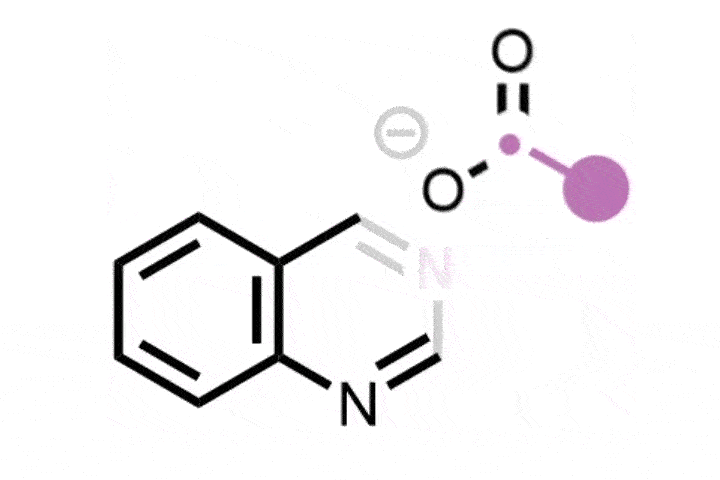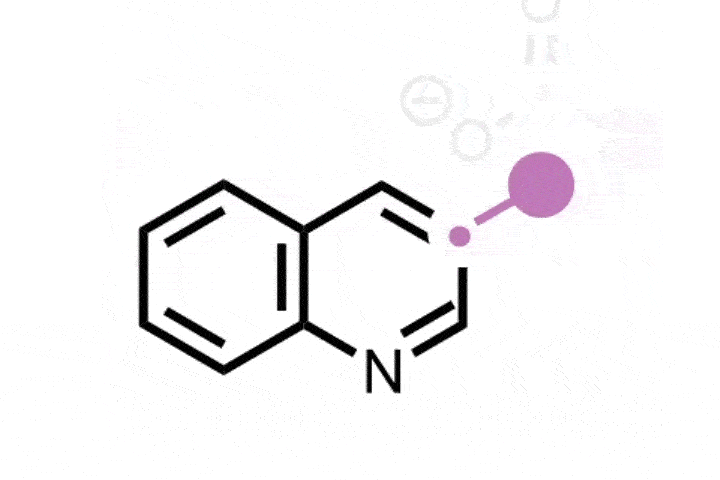
In the reaction, the iodine catalyst first oxidises the amine into an imine. The ring then rearranges by incorporating the imine nitrogen into the ring to generate a nitrilium ion – a protonated nitrile. A methoxy anion from the solvent or the iodine catalyst then attacks the nitrilium to create an imino ether.
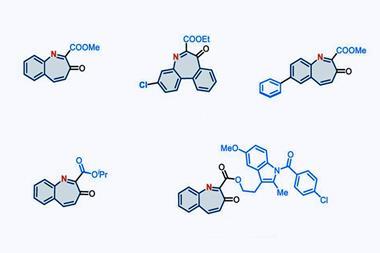
Zhang says that this intermediate reacts with a range of both nucleophiles and electrophiles, including water, azides, alkynes, benzynes, amines and carboxylic acids. This allows the team to synthesise a range of nitrogen heterocycles of different sizes. Depending on the reagent, the ring can expand, contract or simply swap out a carbon atom for a nitrogen.
‘This [work] is a beautiful illustration of taking an abundant compound – amino-bearing cycloalkanes and n-alkanes – and incorporating the nitrogen,’ says Sarpong. He adds that ‘the [team] also report a very impressive carbon deleting amination, which enhances my imagination of what might be possible’.
Zhang’s team are now looking to expand their reaction to functional groups other than amines. He adds that they are also searching for a different reagent that has the same reactivity as their iodine-based compound, as iodine’s large mass means that large-scale synthesis can be difficult to work with.
Intramolecular indole swapping
Chemists have previously developed methods to introduce substituents on the pyrrole ring of an indole, but ‘they often require pre-functionalised substrates, directing groups, strong bases, metal catalysts or carefully tuned conditions’, says Zeng.
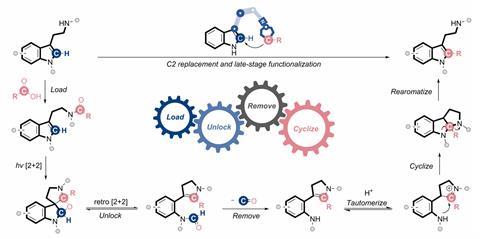
Zeng’s approach instead uses a ‘molecular robotic arm’ to intramolecularly transfer the carbon from an internal carboxylic acid group to the indole’s C2 position.
To do this, the team first loads a carboxylic acid group onto the aminoethyl group of a tryptamine – an indole derivative. Exposing the substrate to UV light causes a cycloaddition reaction that forms three fused rings. These rings then open, lose carbon monoxide and rearrange to regenerate the tryptamine with the carboxylic acid’s carbon incorporated into the pyrrole ring. The team was able to show that this method worked for a range of alkyl, acyl and aryl substituents.
‘This [study] is bold to go after what you might think of as an identity transformation,’ says Mark Levin at the University of Chicago in the US. He adds that replacing one carbon atom with another is ‘a little unusual’.
‘I am actually most excited about the isotopic labelling aspects of the reaction because you can selectively introduce carbon-13, and presumably therefore also carbon-14 radiolabels, to a single position of the indole,’ says Levin. Introducing such isotopes may be useful when studying how indole-based compounds are metabolised inside cells or undergo reactions.
Levin thinks that the major challenge for this method is the need to use a deep UV light source. ‘That limits certain functional groups – [such as] halogens and carbonyls – and also is less commonly used than visible light by most laboratories. I don’t know if the mechanism will translate to other systems, but it’s a very interesting starting point,’ he adds.
Sarpong notes that both reactions ‘accomplish transformations that were the subject of my dreams until now’. ‘In short, these works collectively change the way that I think [about skeletal editing].’
References
1 L Zhang et al, Science, 2026, 392, 512 (DOI: 10.1126/science.aec3587)
2 L-H Li et al, Science, 2026, 392, 528 (DOI: 10.1126/science.aee5416)












No comments yet