Ben Valsler
This week, Kat Arney investigates a compound that can inject a bit of colour into our lives, but not necessarily in a good way…
Kat Arney
What links the following three things: a nasty bruise, faeces, and a common illness in young babies? The answer is bilirubin, a chemical produced from the breakdown of blood, which is responsible for the golden hue of a fading bruise, the brown colour of poop, and the yellowed skin resulting from jaundice.

The journey to making bilirubin starts with haemoglobin – the iron-containing molecule that’s packed into red blood cells and carries oxygen around the body. The average red blood cell doesn’t have a long life – millions of new ones are made every minute in your bone marrow, and each lasts around four months before it’s sent off to the spleen for recycling. Once there, the haemoglobin is broken down into its constituent parts: a protein called globin, and hemin, which is made up of heme – a porphyrin ring – and an ion of iron.
Globins get chopped up into their constituent amino acid building blocks, which go back into the body to make more proteins, and the iron is recycled to make more hemin. But the heme ring can’t be re-used so it is split open, creating biliverdin – a bright green pigment – which is later broken down to produce the yellow-brown compound bilirubin.
The next step happens in the liver, where bilirubin is bound to glycuronic acid in order to make it water soluble. It’s then excreted into the intestine as part of bile – a liquid that enables the body to digest fats. From here, it’s a short hop down the gastrointestinal tract where another reaction takes place, this time engineered by gut bacteria, eventually producing brown-coloured stercobilin which gives faeces its classic poop colour.
Another pigmented breakdown product – yellow urobilin – ends up in urine, putting the gold in your golden shower. Perhaps unsurprisingly, problems with this complex metabolic pathway, such as inherited defects in the enzymes involved or liver disease, can lead to bilirubin building up in the body and causing jaundice, characterised by yellowing of the skin and whites of the eyes.
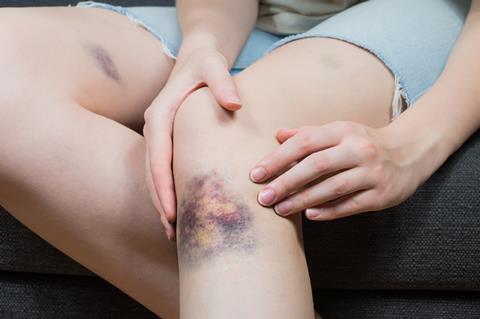
Although biliverdin doesn’t hang around long in the human body, it has a few interesting properties of its own. It’s the chemical that gives bruises a greenish tint after a day or two as fresh blood under the skin clots and gets broken down, eventually fading to browny-yellow as the biliverdin breaks down into bilirubin. It’s also biliverdin that gives blue and green-shelled bird eggs their delicate colours, and the strange green blood of some fish, reptiles and amphibians. It’s also found in high amounts in the placentas of dogs. In fact, there are rare cases of normally yellow or white-coated puppies being born green after receiving an unusually large dose of biliverdin from their mum.
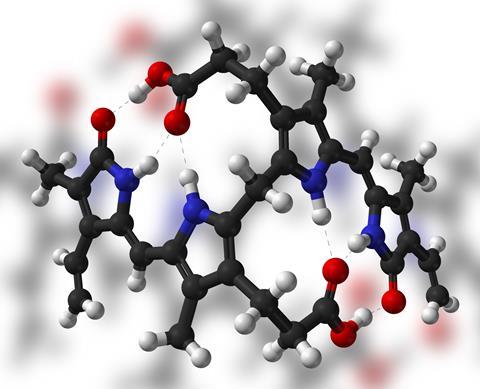
The connection to newborns comes in here too. While they’re in the womb, human fetuses rely on a particular form of haemoglobin known as fetal haemoglobin, which has a very high affinity for oxygen. Once a baby is born, it starts making proper adult haemoglobin and has to break down all the fetal stuff in a hurry, releasing lots of bilirubin into the body. This sudden switch can be overwhelming for an immature liver, especially if the child has been born prematurely, leading to jaundice.
Nearly two thirds of babies will end up with jaundice in the first week or so of their life, although it is often mild and clears up by itself. In the most serious cases, bilirubin can damage the brain and spinal cord, which can be life-threatening. Luckily, there is a simple and lifesaving treatment, which owes its origins to the sharp eyes of Sister Jean Ward, who was in charge of the Premature Baby Unit at Rochford General Hospital in Essex. Back in 1956, Ward noticed that babies that were put out in the sun had less jaundice. At the same time, hospital biochemists were perplexed by the unusually low levels of bilirubin in blood samples that had been left near a sunny window.
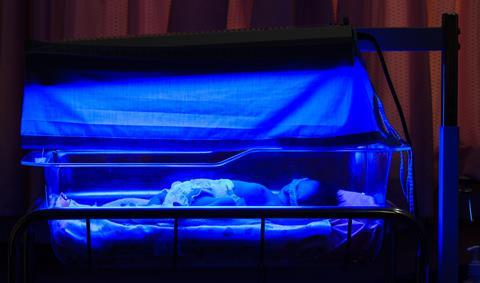
Based on these observations, US paediatrician Jerry Lucey developed a technique called phototherapy, which involves shining a certain wavelength of blue light on the skin of jaundiced babies. This causes a reaction that changes insoluble bilirubin into a water-soluble form, so it can be excreted in urine and faeces. Although Lucey published the results of a randomised controlled trial of the treatment in 1968, the technique took another decade to catch on as many doctors placed their trust in the more invasive but proven approach of blood transfusion. However, the self-styled ‘Prince of Light’ won out over these powers of darkness, and since then phototherapy has saved the lives of countless thousands of newborn babies around the world.
Ben Valsler
That was Kat Arney with bilirubin. Next week, in light of recent political news, I’ll be looking at the collection of minerals that collectively we call asbestos, and how their fantastically useful properties led to widespread use before their devastating health effects came to light, leading to the longest, most expensive mass litigation in American history.
Until then, get in touch with any questions or comments, or compounds you would like us to shine our spotlight on. Email chemistryworld@rsc.org or tweet @chemistryworld. I’m Ben Valsler, thanks for joining me.













No comments yet