Welcome back to what has unexpectedly become the Chemistry in its element coronavirus collection. Seeing as we’ve already looked at two of the four candidate treatments in the World Health Organisation’s Covid-19 Solidarity trial, it seems fitting that we now look at the compounds involved in the remaining two.
Solidarity consists of four arms, but not four compounds: Chloroquine and hydroxychloroquine together make one arm, remdesivir another, and the remaining two arms involve three compounds: the HIV combination lopinavir and ritonavir, and then that same combination but with added interferon beta-1a.
Ritonavir has its own interesting history – which we’ll hear more about next week – so this podcast will focus on lopinavir, and we’ll follow up with interferon later.
Lopinavir is, by Chemistry in its element standards, a pretty new drug. Developed by Abbott Laboratories and patented in the 1990s, it’s been licenced for treatment of HIV since 2000. It’s a small molecule drug and works by blocking the action of an enzyme essential for HIV replication: HIV-1 protease.
If you recall Brian Clegg’s lesson on enzyme language in his podcast on amylase a few weeks ago, you will have already gathered that a protease is an enzyme that breaks down proteins. And although that sounds destructive, in the case of HIV-1 protease, it’s essential for cleaving polyproteins and gathering the building blocks needed to manufacture the infectious virion that carries HIV’s genetic material outside an infected cell. It does the biological equivalent of cutting the right shapes from a sheet of fabric, ready to tailor the virus’ new suit.
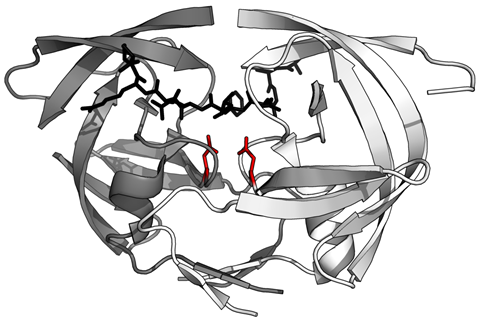
Lopinavir contains a hydroxyethylene scaffold which mimics the sites in polyproteins where HIV protease makes its cuts. But – and if I can slightly over-extend my own metaphor – the protease ‘scissors’ simply can’t get through the lopinavir ‘material’. With all the protease engaged in trying to cleave the uncleavable, the virus is prevented from creating new infectious units, and spread of HIV halts.
Although lopinavir is highly specific for HIV protease, the drug itself is targeted by enzymes produced by the liver – notably the cytochrome P450 3A4 – so it doesn’t last long in the body. Not only does this mean it has to be taken twice daily, but it’s also exclusively administered with small amounts of another drug: Ritonavir. This prevents lopinavir metabolism so it remains in higher concentrations for longer.
Lopinavir may also be effective against the human papilloma virus, or HPV, which can cause cervical cancer . A small trial in a hospital in Nairobi, working with the University of Manchester, offered the treatment to women with high-grade, borderline or low grade disease. Positive results were seen in over 90% of patients. Many countries now offer a preventative HPV vaccine, but as it’s the world’s most common sexually transmitted infection, developing new treatments is always desirable. Lopinavir can even be self-administered as a pessary, a significant bonus in developing countries.

And so to Covid-19. As with other drugs in the Solidarity trial, there is some reason to think that lopinavir and ritonavir may target Sars-CoV-2. Previous studies have shown it effective in improving the outcome for Marmosets infected with the related Mers-CoV, which causes Middle East respiratory syndrome. The World Health Organisation does state that evidence ‘is yet to show it can improve clinical outcomes or prevent infection’, but that ‘there are indications from laboratory experiments that this combination may be effective against Covid-19’. The picture is still quite muddy: results from a Chinese clinical trial published on 18 March this year showed no benefit for hospitalised Covid-19 patients, while in the very same week there were reports of the Moscow City Health Department recommending the lopinavir & ritonavir combination for milder cases. As the results from the global trial start to come in, a picture should start to develop showing which, if any, of these drugs can stop the pandemic in its tracks.
Next week, Alexander Whiteside takes the other half of this combination treatment, with a cautionary tale of polymorphism & seeding.
Alexander Whiteside
It’s not clear where Form Two came from. But once Form Two existed, it carried its own template, for a new, less useful version of ritonavir everywhere it went. And Form Two went everywhere.
Ben Valsler
In the meantime, find all of our Covid-19 content – available for free – at chemistryworld.com/coronavirus, and all of our previous podcasts at chemistryworld.com/podcasts. And please do reach out with any questions, comments or compound suggestions; we’re at chemistryworld@rsc.org and on twitter we’re @chemistryworld. I’m Ben Valsler, thank you for joining me.
Additional information
Theme: Opifex by Isaac Joel, via Soundstripe
Additional music: Currents and Help and hope by Marie, via Soundstripe











1 Reader's comment