Researchers have experimentally proved that the simplest Diels–Alder reaction between 1,3-butadiene and mono-substituted alkenic dienophiles is not endo-selective as previously thought. Mick Sherburn, who led the work at the Australian National University, says many chemists will be surprised by their findings, and hopes they change the way that people think about the reaction and teach it.
The Diels–Alder reaction is one of the most important reactions in organic chemistry, and earned Otto Diels and Kurt Alder the Nobel prize in Chemistry in 1950. The [4+2] cycloaddition between a conjugated diene and a substituted alkene is a powerful synthetic tool, forming substituted cyclohexene derivatives with up to four new stereocentres. Although the endo/exo selectivity of the diastereomeric products formed from substituted olefinic dienophiles has been extensively researched, it is still not fully understood.
Initial studies by Alder and William Stein shaped the Alder endo rule, which states that Diels–Alder reactions preferentially undergo the endo mode of addition. However, an increasing number of counter-examples bring this rule under scrutiny. Additionally, current studies use a limited range of alkene dienophiles and so do not clearly show whether endo-selective 1,3-butadiene reactions are more generally preferred.
Now, Sherburn and his colleagues have conclusively demonstrated that simple Diels–Alder reactions are not highly endo-selective. ‘Experimentally, endo/exo-selectivity in a Diels–Alder reaction of 1,3-butadiene is cryptic: It’s not possible to determine whether the endo- or exo-pathway has been followed with unlabelled 1,3-butadiene, since both pathways give the same product,’ explains Sherburn.
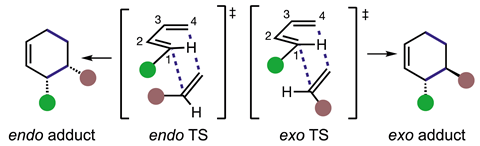
To overcome this, the team stereoselectively synthesised (1E,3E)-1,4-dideutero-1,3-butadiene to investigate the products formed. Collaborating with Michael Paddon-Row at the University of New South Wales, the team investigated the reaction between dideuterated 1,3-butadiene and 10 common dienophiles and found commonly used mono-substituted alkenic dienophiles are not highly endo-selective in thermal Diels–Alder reactions. ‘Figuring out how to make and use this [deuterium-labelled 1,3-butadiene] took a lot of skill and perseverance,’ Sherburn says, in part due to its boiling point of –4°C. The team further confirmed their experimental results theoretically using quantum chemical calculations to determine the endo:exo product ratios.
Nick Greeves, an organic chemist at University of Liverpool, UK, and co-author of the classic undergraduate textbook Organic Chemistry, is not convinced the high endo-selectivity perception is widespread. ‘I think that people would expect reactions with only a single electron withdrawing group to be slightly endo-selective, some more than others. And indeed, that’s what they demonstrated,’ he says. ‘We’ve discovered that some of these reactions are a bit endo-selective, but 60:40 isn’t no selectivity.’ Sherburn says it is reasonable to call the reactions non-stereoselective: ‘The most important point is that the energy difference between [the] endo- and exo-pathways is negligible.’
However, Greeves suggests the computational approach could predict the selectivity of other systems. ‘It isn’t easy to replicate ratios of diastereoisomers based on the small difference in energy, and they’ve done a nice job with that. Given the ubiquity of the Diels–Alder reaction, that could be extremely useful,’ he says.
Sherburn plans to use their computational method to pinpoint the best synthetic approaches of structurally complex molecules to invent better total syntheses using Diels–Alder reaction sequences.



















No comments yet