Organic chemistry – Page 41
-
 Research
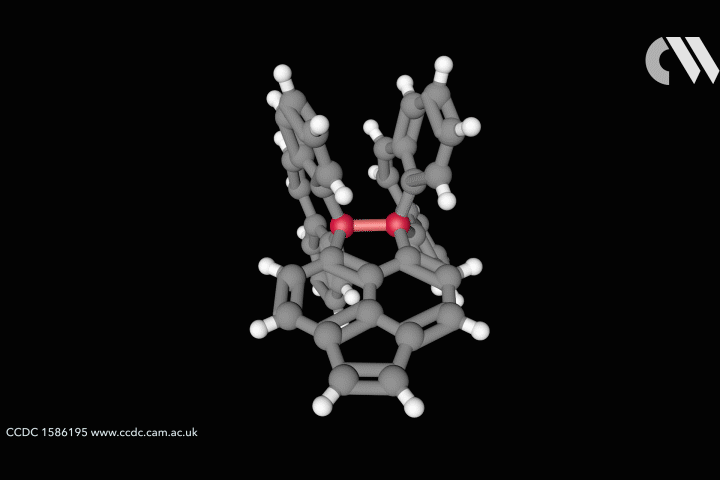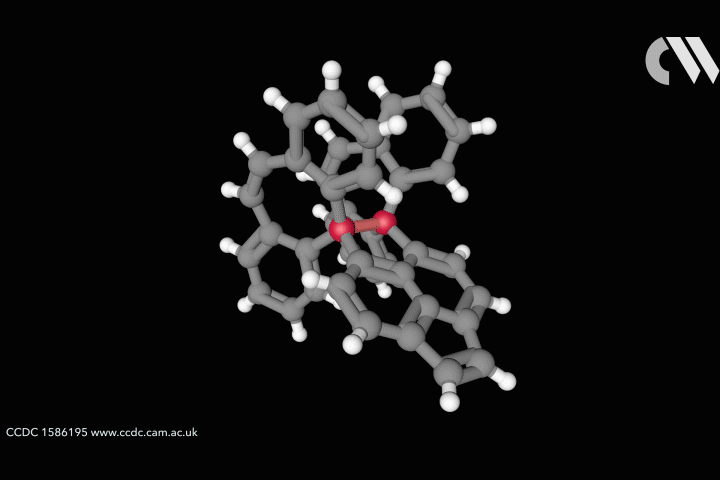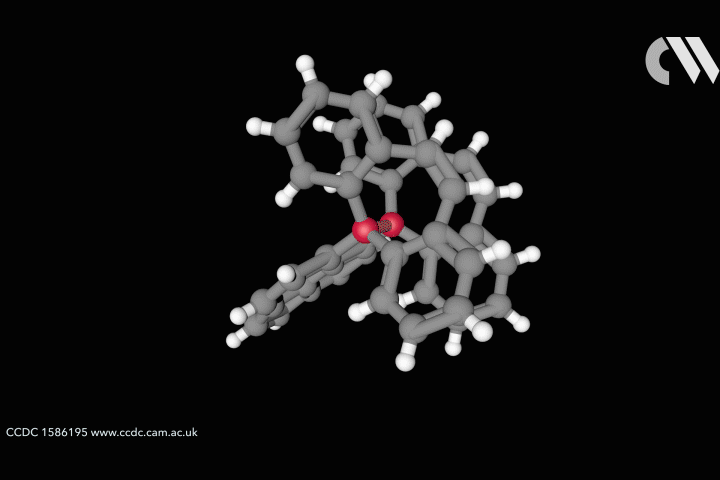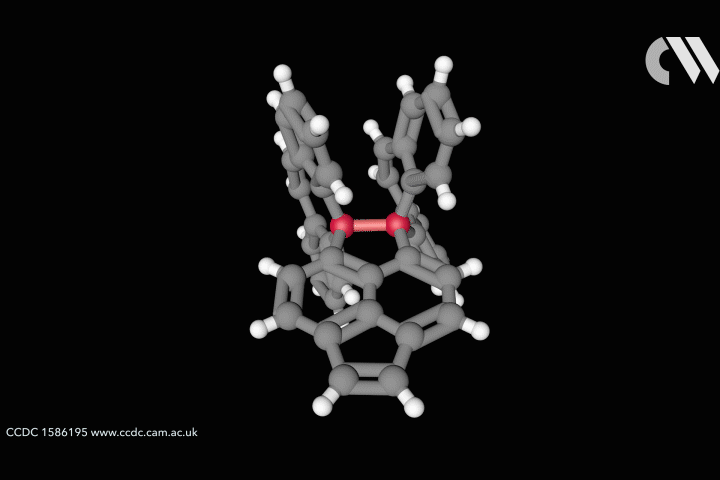
ResearchLongest carbon–carbon bond yet pushes chemistry to its limits
Steric strain creates bond longer than theory thought possible
-
 Article
ArticleIn conversation with Robert Grubbs, Nobel laureate
Following the recent acquisition of the world-leading Materia catalyst business by Umicore, we’ve been granted an exclusive interview with Robert Grubbs, chemistry Nobel laureate and co-founder of Materia.
-

-
 Research
ResearchSimple synthesis makes curious chiral clamshell molecule
Compounds with two chiral axes hold promise for catalysis and even molecular machines
-
-
 Research
ResearchAmerican Civil War era tea yields modern day medicine
Macrocyclisation strategy key to obtaining long known alkaloid with untapped potential
-
 Research
ResearchAI-invented syntheses are a hit in their lab debut
First proof that routes devised by an algorithm work in practice could make organic chemistry accessible to non-experts
-

-
 Research
ResearchAntimalarial plant's chlorophyll catalyses drug synthesis
New ‘green’ method makes malaria-fighting artemisinin’s synthesis faster and cheaper with industrial production planned for 2021
-
 Research
ResearchEncryption using carbon-13
New technique encrypts materials by altering levels of carbon-13 at regiospecific atoms
-
 Research
ResearchMechanochemistry first as reaction driven by pressure
A redox reaction triggered by squeezing a crystal results in the ejection of copper nanoparticles
-
 Research
ResearchSix degrees of chemical separation
Network analysis provides insight into navigating chemical space
-
 Research
ResearchMachine learning triumphs in tough cross coupling challenge
5000 nanoscale experiments teach algorithm how to predict outcomes of reactions in the presence of inhibitors
-

-
-

-
 Research
ResearchCarbyne equivalents fire up carbon–carbon bond formation
‘Easy-to-make’ reagent adds a wide variety of functional groups to aromatic rings
-
 Research
ResearchUnpicking a unique molecular network
Computational modelling informs self-assembly solution
-
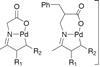 Opinion
OpinionKetones perform at the palladium
C–H activation takes the stress out of organometallic couplings
-
 News
NewsWanted: synthetic chemists (humans need not apply)
Automation could free chemists from tedious lab work – if they’re ready to think differently about research