Simple optical lens behind strategy for manipulating photochemical reactivity
Researchers in Switzerland have shown that both available redox potential and final product selectivity is controllable with a light intensity-sensitive photocatalyst.
Photocatalysts have many promising applications, including removing pollutants from wastewater, storing energy in the form of solar fuels and organic synthesis. As each type of photoreaction has one optimal catalyst, several expensive photocatalysts are often required to create the desired product. Creating catalysts with properties that can be controlled and altered with light could allow a single catalyst to work on several reactions, reducing cost.
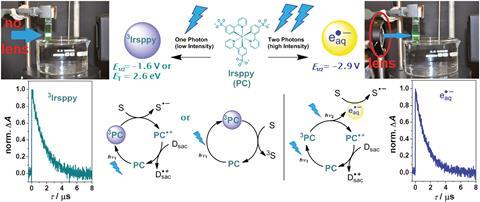
Light wavelength can influence the redox potential of some catalysts, but precise wavelengths require expensive light sources. Christoph Kerzig and Oliver Wenger at the University of Basel, Switzerland, have instead turned their attention to light intensity. They show they can control the reaction of a photocatalyst, solvent and sacrificial reagent using a lens to apply different light intensities.
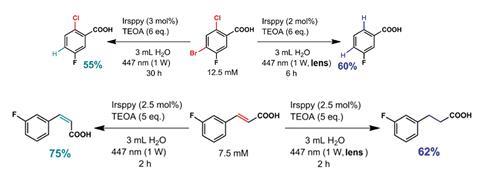
By irratidating an iridium catalyst, Irsppy, with one photon of light using a continuous wave laser, the team demonstrated that the excited triplet state, 3Irsppy, is capable of catalysing debromination reactions. Increasing the light intensity with a lens delivers two photons of light within the triplet state’s lifetime, exciting 3Irsppy further to generate a solvated electron, an aggressive super-reductant. The solvated electron almost doubles the redox potential available allowing challenging dechlorination reactions to occur.
‘Importantly, the two-photon strategy can even activate inert substrates, whose activation is impossible with one visible photon,’ Kerzig says. The team went on to see if their strategy would work with substrates that activate via different mechanisms. They demonstrate that trans-3-fluorocinnamate undergoes double bond isomerisation in the presence of Irsppy, but with the lens the reaction becomes a hydrogenation providing a different product.
‘Our approach could permit novel cascade-type photochemistry with the light intensity as the selectivity parameter. For the latter, reaction sequences with isomerisation, hydrogenation, cycloaddition and arylation steps are conceivable,’ adds Wenger.
Organic chemist Dennis Cao, of Macalester College in the US, describes the idea as novel. ‘This proof of principle is sure to inspire the development of new small molecule and polymer synthesis methodologies that exploit this switchable reactivity to achieve otherwise challenging synthetic connections.’
References
This article is open access
C Kerzig and O S Wenger, Chem. Sci., 2018, DOI: 10.1039/c9sc04584h
















No comments yet